Functional Dark Matter — Experimentally Prioritized Novel Genetic Systems
CompletedResearch Question
Which genes of unknown function across 48 bacteria have strong fitness phenotypes, and can biogeographic patterns, pathway gap analysis, and cross-organism fitness concordance — combined with existing function predictions and conservation data — prioritize them for experimental follow-up?
Research Plan
Hypothesis
- H0: Genes annotated as "hypothetical" or "uncharacterized" with strong fitness effects are functionally random — their fitness phenotypes, conservation patterns, and environmental distributions are indistinguishable from annotated genes.
- H1: Functional dark matter genes form coherent groups: they cluster into co-regulated modules with annotated genes, show non-random phylogenetic distributions, and their pangenome conservation and biogeographic patterns correlate with the environmental conditions under which they show fitness effects.
Sub-hypotheses
- H1a (Functional coherence): Dark genes with strong fitness effects are enriched in ICA fitness modules alongside annotated genes, enabling guilt-by-association function prediction. (Partially addressed by
fitness_modules— this project quantifies coverage and tests on the full dark gene set.) - H1b (Conservation signal): Dark genes important under stress conditions are more likely to be accessory (environment-specific) than dark genes important for carbon/nitrogen metabolism (which should be more core).
- H1c (Cross-organism concordance): Dark gene families with orthologs in multiple FB organisms show concordant fitness phenotypes across organisms (same condition classes cause fitness defects). The pre-computed
specogtable provides per-OG condition-level fitness summaries. - H1d (Biogeographic pattern): For accessory dark genes, genomes carrying the gene come from environments that match the lab conditions where the gene shows fitness effects (e.g., genes important under metal stress are enriched in genomes from contaminated sites).
- H1e (Pathway integration): Some dark genes fill "steps_missing" gaps in otherwise-complete GapMind pathways, suggesting they encode novel enzymes for known metabolic functions. GapMind
score_categoryuses a four-level hierarchy:likely_complete>steps_missing_low>steps_missing_medium>not_present; multiple rows per genome-pathway pair must be aggregated (MAX score) before interpreting gaps.
Approach
Phase 1: Integration & Census (NB01)
Goal: Build a unified dark gene table by loading and merging all existing data products, then characterizing the full dark gene landscape.
What is loaded (not re-derived):
1. All FB genes (gene table, 228K) — classify annotation status:
- Hypothetical: desc contains "hypothetical protein" or is empty/null
- DUF: desc contains "DUF" (domain of unknown function)
- Uncharacterized: desc contains "uncharacterized" or "unknown"
- Partial: has domain hits (PFam/TIGRFam in genedomain) but no full functional annotation
- Annotated: clear functional description (control group)
2. Essential gene classification from essential_genome: load orphan essentials (7,084 genes, 58.7% hypothetical) as a separate dark matter class. These have zero rows in genefitness (no viable mutants) and would be missed by fitness-effect filters alone.
3. Fitness phenotype summary per dark gene: from specificphenotype (38K entries) and genefitness (conditions where |fit| > 1 and |t| > 3, with CAST to float)
4. Condition-class characterization: join genefitness → experiment to tag each fitness effect with its experiment group (stress, carbon source, nitrogen source, etc.) — this is the foundation for lab-field concordance in NB04
5. Pangenome conservation from fb_pangenome_link.tsv (177,863 links): core/accessory/singleton status per gene
6. Module membership from fitness_modules project: which ICA module each gene belongs to, and the module's existing function prediction (6,691 predictions)
7. Ortholog families from essential_genome project: 17,222 ortholog groups, essentiality classification
8. Co-fitness top partners from cofit table: for dark genes NOT in any ICA module, load top 5 co-fitness partners with their annotations — provides functional context for the ~40% of dark genes outside modules
9. Domain annotations from genedomain: PFam, TIGRFam, CDD hits that provide partial structural clues even for "hypothetical" genes
Output: data/dark_genes_integrated.tsv — unified table (~54K dark genes, ~3,700 with strong fitness effects, plus ~4,100 hypothetical essentials) with all cross-references.
Phase 2: New Inference Layers (NB02)
Goal: Apply inference methods not covered by prior projects.
- GapMind pathway gap-filling:
- For the ~44 FB-linked species, query
gapmind_pathwaysfiltered by those species' genome IDs (not a full 305M-row scan) - Aggregate multiple rows per genome-pathway pair using MAX score (per the
score_categoryhierarchy:likely_complete>steps_missing_low>steps_missing_medium>not_present) - Use two-stage aggregation: genome-pathway → species-pathway
- Identify pathways scored as
steps_missing_low(nearly complete) and check if dark genes from those species could fill the missing enzymatic steps -
Match via EC numbers, KEGG reactions, or PFAM domains between dark gene annotations and missing pathway steps
-
Cross-organism fitness concordance:
- For dark gene ortholog families (from
essential_genomeortholog groups) present in 3+ FB organisms, build a "family fitness fingerprint": condition-class × organism matrix of fitness effects - Use the pre-computed
specogtable (specific phenotypes by ortholog group) which already hasminFit/maxFit/minT/maxTper OG × condition — avoids re-querying 27M genefitness rows - Test concordance: do orthologs of the same dark gene show fitness effects under the same condition classes?
-
Concordant families get higher confidence for functional inference
-
Pangenome-wide phylogenetic distribution:
- For dark gene families, extract eggNOG OG identifiers from
eggnog_mapper_annotations(viafb_pangenome_link→gene_cluster_id→query_name) - Create a temp view of target OG IDs and JOIN against
eggnog_mapper_annotationsto find all clusters across 27,690 species sharing the same OG - Map to taxonomy via
gene_cluster.gtdb_species_clade_id→gtdb_species_clade→gtdb_taxonomy_r214v1 - Compute phylogenetic breadth: number of phyla, orders, families, species carrying each dark gene family
- Classify as: clade-restricted (one phylum) vs widespread (3+ phyla)
Output: data/gapmind_gap_candidates.tsv, data/concordance_scores.tsv, data/phylogenetic_breadth.tsv
Phase 3: Biogeographic Analysis (NB03)
Goal: Determine whether the environmental distribution of dark gene carriers shows non-random patterns, and whether those patterns match lab fitness conditions.
- Geographic distribution of carriers:
- For each dark gene family (from NB02 phylogenetic distribution), identify all pangenome species carrying related clusters
-
For genomes in those species, extract:
- AlphaEarth embeddings (83K genomes, 28% coverage — safe to collect via
.toPandas()) - NCBI isolation source categories from
ncbi_env(EAV format — pivot byharmonized_namefor key attributes) - Geographic coordinates from
gtdb_metadata.ncbi_lat_lon
- AlphaEarth embeddings (83K genomes, 28% coverage — safe to collect via
-
Within-species carrier vs non-carrier test (strongest test — controls for phylogeny):
- For accessory dark genes, within each species that has the gene in some but not all genomes:
- Compare AlphaEarth embedding distributions between carriers and non-carriers
- Compare NCBI isolation source category frequencies
- Use permutation tests or Mann-Whitney U for significance
-
Report N genomes with metadata / N total for every comparison
-
Environmental characterization summary:
- Cluster carrier genomes by AlphaEarth embedding space (UMAP + HDBSCAN)
- Summarize NCBI isolation source categories per dark gene family
- Flag dark gene families with strong environmental signals
Output: data/biogeographic_profiles.tsv, data/carrier_noncarrier_tests.tsv
Phase 4: Lab-Field Concordance & NMDC Validation (NB04)
Goal: Test whether dark genes' lab fitness conditions match the environmental contexts where they appear in nature.
- Pre-registered condition-environment mapping (define BEFORE looking at biogeographic data):
| FB Experiment Group | Environmental Category | NCBI/AlphaEarth Signal |
|---|---|---|
| Metal stress (Cr, Ni, Zn, U, Cu) | Contaminated sites | ncbi_env "contaminated", lat/lon near industrial sites |
| Carbon source utilization | Carbon-rich environments | ncbi_env "soil", "sediment", DOC-rich |
| Nitrogen source | Nitrogen-variable environments | ncbi_env "agricultural", nitrogen-related |
| Osmotic/salt stress | Marine/saline | ncbi_env "marine", "saline", "brackish" |
| Temperature stress | Extreme thermal | ncbi_env "hot spring", "permafrost" |
| pH stress | pH-extreme environments | ncbi_env "acidic", "alkaline" |
| Oxidative stress | Oxic/anoxic transitions | ncbi_env "sediment", redox gradients |
- Concordance test: For each dark gene family with both (a) condition-specific fitness effects and (b) environmental metadata for carriers:
- Is the carrier environmental profile enriched for the predicted environment category?
-
Fisher's exact test per family, BH-FDR correction across all families
-
NMDC independent validation (supplementary — community-level, not gene-level):
- For taxa carrying dark genes, check abundance in NMDC
taxonomy_features(6,365 samples, 3,493 taxa — CLR-transformed wide matrix; resolve taxon IDs viataxonomy_dim/taxstring_lookup) - Correlate with NMDC
abiotic_features(22 environmental measurements per sample) - Use NMDC
trait_features(92 community-level functional traits) for functional context - Frame as "consistent with" or "independently corroborated by" — never causal
- Report concordance rate and note genus-level resolution limitation
Output: data/lab_field_concordance.tsv, data/nmdc_validation.tsv
Phase 5: Prioritization & Synthesis (NB05)
Goal: Produce a ranked candidate list for experimental characterization.
Scoring dimensions (each 0-1, weighted):
1. Fitness importance (0.25): max |fitness| across conditions, number of specific phenotypes, essentiality status
2. Cross-organism conservation (0.20): number of FB organisms with ortholog, fitness concordance score from NB02
3. Functional inference quality (0.20): module membership with prediction, co-fitness with annotated genes, domain clues, GapMind gap-filling evidence
4. Pangenome distribution (0.15): phylogenetic breadth (from NB02), core vs accessory classification
5. Biogeographic signal (0.10): strength of environmental pattern (from NB03), lab-field concordance score (from NB04)
6. Experimental tractability (0.10): in genetically tractable FB organism, not essential (can knock out), has characterized co-fitness partners
Output: Top 50-100 candidates with:
- Gene IDs across all organisms carrying orthologs
- Best functional hypothesis with evidence type and confidence
- Suggested experimental approach (which organism, which conditions)
- Environmental context summary
- All prior project cross-references (module ID, ortholog family, conservation class)
Revision History
- v1 (2026-02-25): Initial plan
- v2 (2026-02-25): Restructured to build explicitly on prior projects (
fitness_modules,essential_genome,conservation_vs_fitness,module_conservation). Reduced from 6 to 5 notebooks by consolidating census + integration. Incorporated plan review feedback: essential genes as separate dark class, GapMind score hierarchy,specogtable for concordance, concrete pangenome-wide query strategy, Spark session pattern. - v3 (2026-02-26): Added NB08 (conserved gene neighborhoods, cofit-validated operons, improved prioritization). Cross-species synteny validation of NB07 operon predictions using ortholog group neighborhood conservation across 48 FB organisms. Cofit validation of non-essential operon pairs via tiered scoring (mutual top-5 through one-directional top-20). Re-scored all fitness-active (17,344) and essential (9,557) dark genes with new evidence. Evidence-weighted experimental roadmap incorporating synteny, cofit, and phylogenetic gap signals. Added Finding 12 to REPORT.md and updated limitations to explicitly compare methodology against DOOR/STRING/EFI-GNT standards.
- v4 (2026-02-26): Added NB09 (final synthesis). Darkness spectrum classifies all 57,011 dark genes into 5 tiers (T1 Void through T5 Dawn) based on 6 binary evidence flags. Greedy weighted set-cover selects organisms covering 95% of priority value; MR-1 ranks first. Per-organism experimental action plans classify genes as hypothesis-bearing (specific condition recommendations) vs. darkest (broad screen). Added Finding 13 to REPORT.md, updated data/figure/notebook tables.
- v5 (2026-02-26): Updated Analysis Plan to describe all 9 notebooks (NB06–NB09 were missing). Added NB06 (robustness checks), NB07 (essential gene prioritization), NB08 (cross-species synteny + cofit validation), NB09 (final synthesis) with goals, outputs, and figures. Rewrote REPORT.md Results section with narrative rationale explaining why each analysis step was needed and how it builds on the prior step. Added Experimental Recommendations section to REPORT.md as the front-and-center deliverable: top candidates with evidence rationale, top 5 organisms with selection justification, and a three-experiment starting campaign.
- v7 (2026-02-27): Added NB10 (review improvements). Addresses 2 critical and 4 important suggestions from automated review: (1) Gene-to-gap enzymatic domain matching for GapMind candidates (42,239 matches across 3,186 dark genes, 5,398 high-confidence EC matches). (2) Robust rank indicators showing 18 fitness-active and 6 essential genes remain always-top-50 across all weight configurations. (3) Species-count scoring variant (Spearman ρ=0.982 vs original, 62% top-50 overlap). (4) NMDC sign tests (7/7 trait p=0.0078; 4/4 abiotic p=0.0625) and compositional inflation factor (~20×). (5) Biogeographic binomial test (29/47, p=0.072) and Fisher's combined (p=0.031). All analyses in a single supplementary notebook using pandas/scipy only (no Spark). REPORT.md updated with new findings, limitations, and tables.
- v8 (2026-02-27): Added NB11 (conservation × dark matter classes). Queries full pangenome (27,690 species) for taxonomic breadth of dark gene OGs, replacing the coarse eggNOG breadth_class. 8-tier taxonomic classification (kingdom → species + mobile) with mobile element detection via COG-X and phylogenetic patchiness. 3-tier hypothesis status (strong hypothesis, weak lead, true knowledge gap). Importance score = conservation × ignorance ranks OGs for experimental prioritization. Conservation-weighted minimum covering set orders organisms for maximum novel functional discovery. Per-organism experimental plans with tier × hypothesis breakdowns. Added Finding 14 to REPORT.md, updated data/figure/notebook tables.
- v9 (2026-02-27): NB11b extends conservation to full pangenome. NB11's original Spark query only searched within the 48 FB organism gene clusters (species counts bounded at 33); NB11b's corrected query explodes eggNOG_OGs across all 93.5M pangenome annotations, finding every gene cluster in 27,690 species that shares our root_ogs. Species counts now range 1–27,482 (median 135). OG_id propagation recovers 5,206 additional dark genes (57.5% → 66.6% coverage). Tier distribution flips from 3.5% kingdom to 55.9% kingdom — over half of dark gene OGs are pan-bacterial. Extended tractable organisms file (73 organisms: 48 FB + 25 literature-curated) prepared for future covering set expansion beyond FB. All downstream analyses (importance scoring, covering set, experimental plans) re-executed with corrected conservation data.
- v6 (2026-02-27): Fixed 3 critical bugs and 1 moderate issue from automated review. (1) NB05
score_pangenome()breadth_class vocabulary mismatch — scoring function checked for'widespread'/'clade-restricted'strings that never existed in data (NB02 produces'universal'/'pan-bacterial'/'multi-phylum'/'narrow'); phylogenetic breadth sub-score was silently zeroed for all 17,344 genes. Fixed to match NB02 vocabulary. Score range shifted from 0.048–0.650 to 0.048–0.715. (2) NB09ORG_GENUShardcoded dict mapped only ~21 of 48 organisms; replaced with load fromorganism_mapping.tsv(covers 44/48) plus 4 manual fallbacks. Covering set now 42 organisms (28 genera) with correct genus-redundancy penalties. (3) Addedumap-learn>=0.5torequirements.txt. (4) Removedmatplotlib.use('Agg')and changedplt.close()toplt.show()in NB03–NB09 for inline figure rendering. All 7 notebooks re-executed; REPORT.md updated with corrected numbers. - v10 (2026-02-27): Dual-route framework and extended covering set (NB11c). Formalized two complementary analytical routes: Route A (NB05–NB09, evidence-weighted) optimizes for genes with testable hypotheses; Route B (NB11/11b/11c, conservation-weighted) optimizes for discovering functions of broadly conserved unknowns. NB11c uses Spark to query genus-level OG membership for 25 non-FB organisms (from
extended_tractable_organisms.tsv) across the full pangenome, then runs the conservation-weighted covering set with 73 candidate organisms. Adds organisms from Bacillota, Actinomycetota, and Campylobacterota — phyla absent from the 48-organism Fitness Browser. REPORT.md restructured: Experimental Recommendations now presents both routes with route comparison table; new Limitation #12 (Proteobacteria bias); new Future Direction #6 (expanded organism set); Step 9 added to Results for NB11 analysis.
Overview
Nearly one in four bacterial genes lacks functional annotation ("hypothetical protein"), yet many have experimentally measured fitness effects in the Fitness Browser's 27M measurements across 7,552 conditions. Previous observatory projects have already: predicted function for 6,691 hypothetical proteins via ICA modules (fitness_modules), identified 1,382 hypothetical essentials (essential_genome), and linked 177,863 FB genes to pangenome conservation status (conservation_vs_fitness).
This project builds on those foundations with genuinely new analyses: (1) GapMind pathway gap-filling to find dark genes encoding missing enzymatic steps, (2) cross-organism fitness concordance testing whether orthologs of the same dark gene show the same phenotypes, (3) biogeographic analysis using AlphaEarth satellite embeddings and NCBI metadata to test whether carrier environments match lab fitness conditions, and (4) integrated experimental prioritization producing a ranked candidate list.
Key Findings
Finding 1: One in four bacterial genes is functionally dark, and 17,344 have experimentally measurable phenotypes
Across 48 Fitness Browser organisms (228,709 genes), 57,011 (24.9%) lack functional annotation ("hypothetical protein," DUF, or "uncharacterized"). Of these, 7,787 show strong fitness effects (|fitness| ≥ 2 in at least one condition), and 9,557 are essential (no viable transposon mutants). Together, these 17,344 genes represent the experimentally actionable "dark matter" — genes with clear biological importance but unknown function.
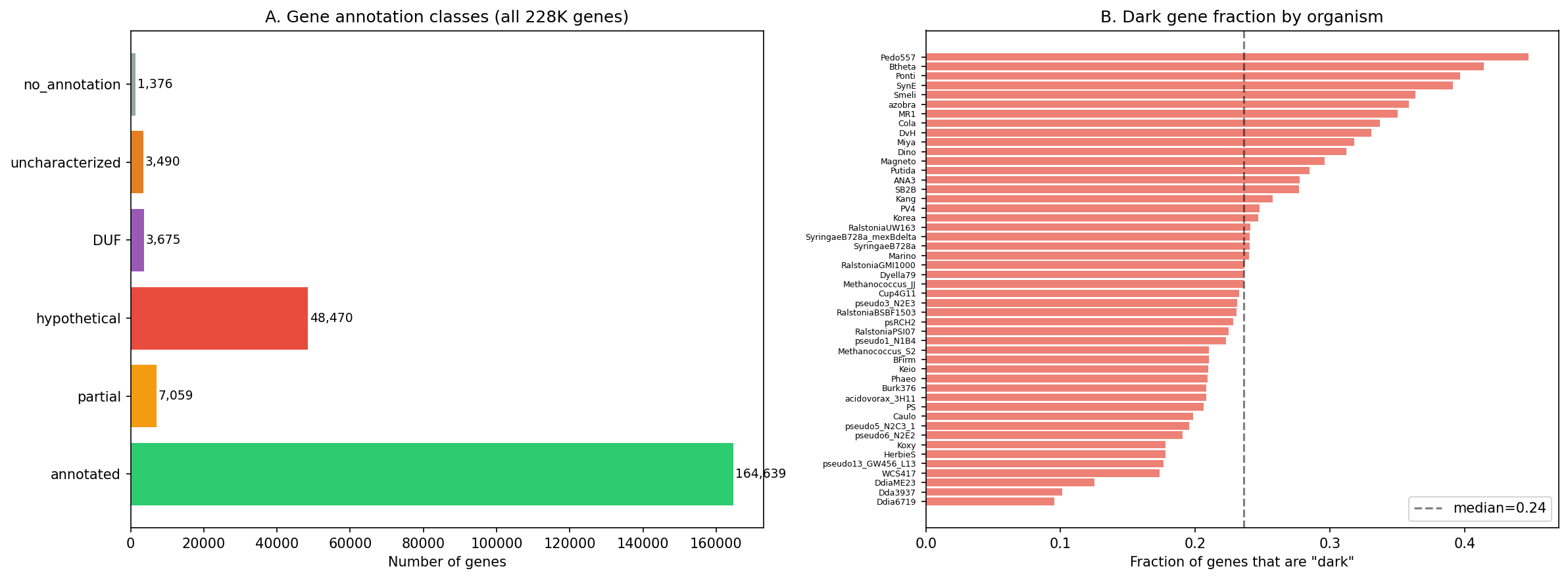
Dark genes are not randomly distributed across organisms: some species have >35% hypothetical genes while others have <15%, reflecting differences in annotation depth rather than true functional content.
(Notebook: 01_integration_census.ipynb)
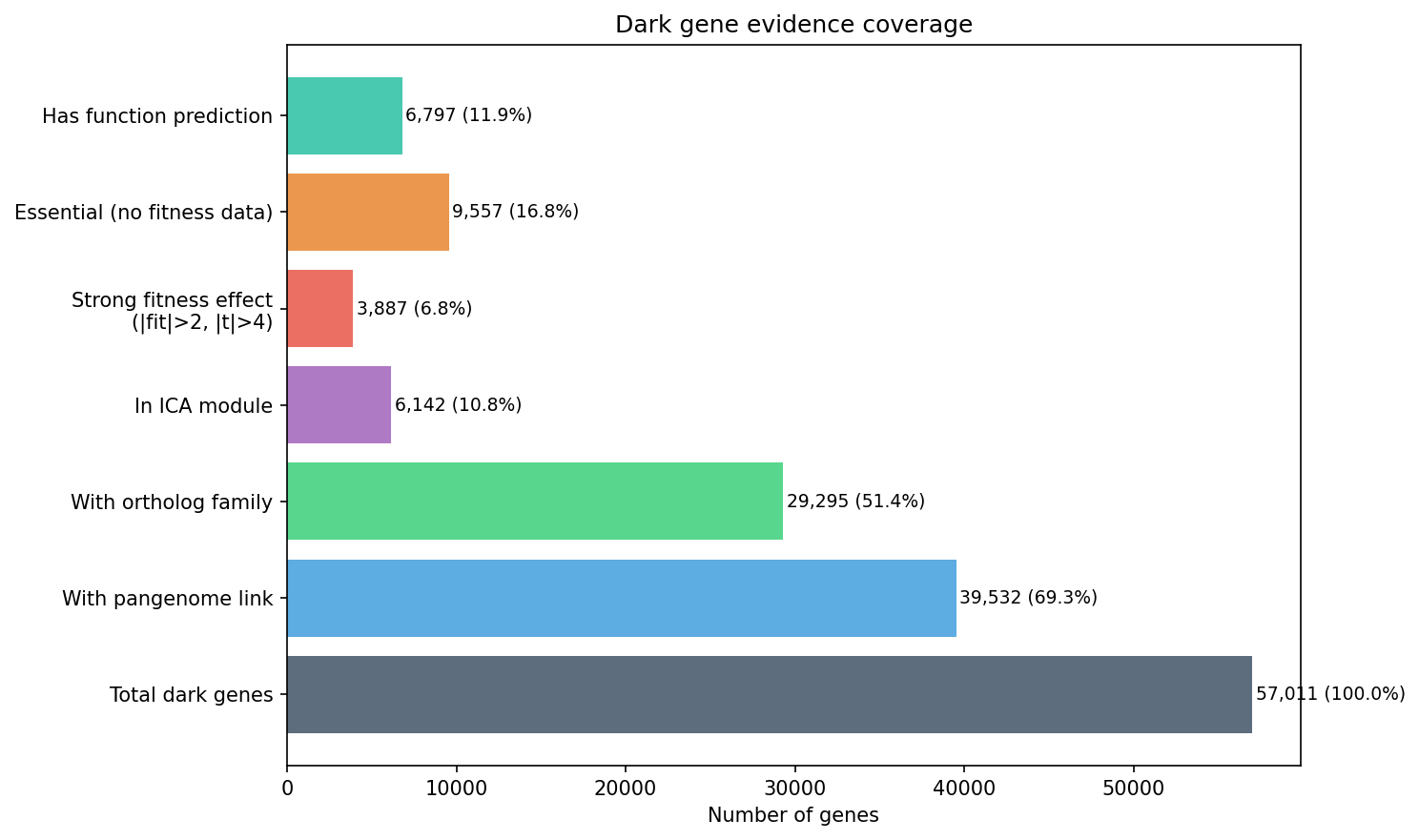
Finding 2: 39,532 dark genes link to the pangenome; 6,142 belong to co-regulated fitness modules
Of 57,011 dark genes, 39,532 (69.3%) have pangenome links via the conservation_vs_fitness project. Among these, 12,686 are accessory (environment-specific) and 511 are both accessory and have strong fitness effects — the prime candidates for biogeographic analysis. Additionally, 6,142 dark genes belong to ICA fitness modules from the fitness_modules project, providing guilt-by-association function predictions.
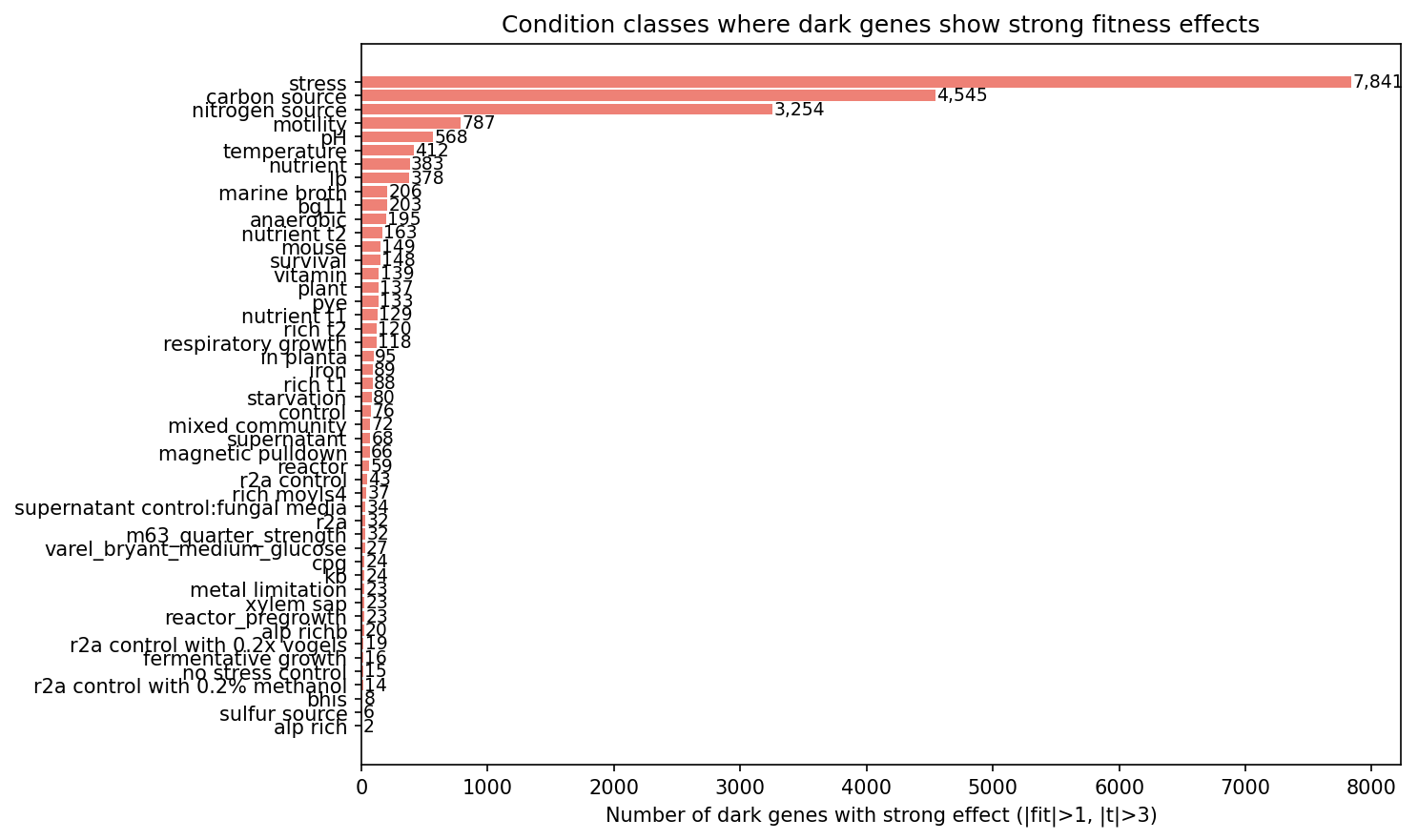
Stress conditions (metals, oxidative, osmotic) dominate among dark genes with strong fitness effects, followed by carbon source utilization and nitrogen source utilization.
(Notebook: 01_integration_census.ipynb)
Finding 3: GapMind identifies 1,256 organism-pathway pairs with metabolic gaps in species harboring dark genes
Across 44 FB-linked species, GapMind pathway analysis identified 1,256 organism-pathway pairs with nearly-complete metabolic pathways (score: steps_missing_low) where dark genes with strong fitness effects co-occur. Note: These are organism-level co-occurrences — each pair represents a species that has a nearly-complete pathway AND harbors dark genes, but no direct gene-to-gap enzymatic matching was performed. The co-occurrence suggests dark genes could encode missing steps, but confirming this requires EC number matching, structure prediction, or experimental validation. The most frequently gapped pathways are carbon source utilization pathways:
| Pathway | Category | Organisms with gaps | Example organisms |
|---|---|---|---|
| Fucose utilization | carbon | 32 | Marinobacter, P. stutzeri RCH2, D. vulgaris |
| Rhamnose utilization | carbon | 31 | Marinobacter, P. putida, Phaeo |
| Sorbitol utilization | carbon | 30 | D. desulfuricans, D. vulgaris, Miyama |
| Myoinositol utilization | carbon | 28 | P. putida, P. syringae, WCS417 |
| Gluconate utilization | carbon | 26 | Marinobacter, D. desulfuricans |
| Asparagine biosynthesis | amino acid | 24 | across diverse phyla |
The organisms with the most gapped pathways — Marinobacter (49), D. desulfuricans ME-23 (45), P. stutzeri (45) — are also those with the largest dark gene complements and most specific fitness phenotypes, suggesting that their "missing steps" may be encoded by functionally dark genes.
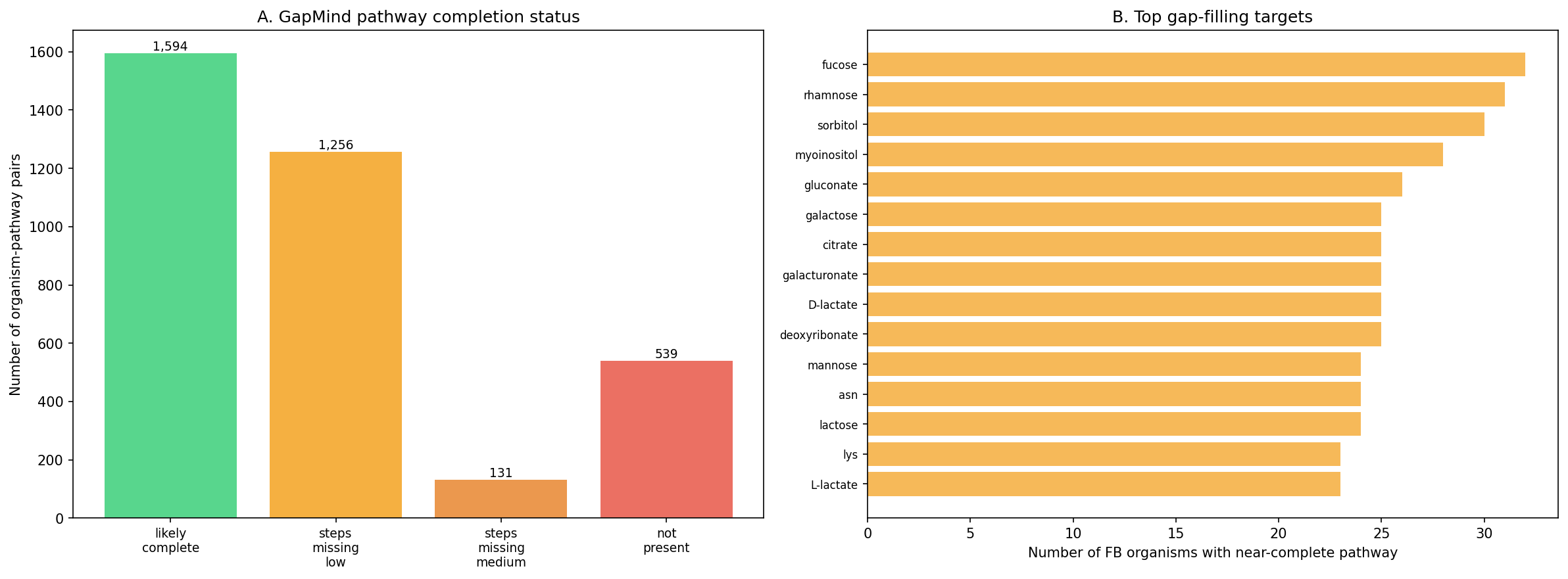
Supplementary domain matching (NB10 Section 1): To move beyond organism-level co-occurrence, curated pathway-enzyme mappings (EC prefixes, PFam families, functional keywords) were used to identify dark genes with annotations compatible with gapped pathways. Of 1,256 organism-pathway pairs, domain matching identified 42,239 gene-pathway candidates across 3,186 unique dark genes, with 5,398 high-confidence (EC prefix match), 4,687 medium-confidence (PFam family match), and 32,154 low-confidence (keyword match) hits. These domain-compatible candidates narrow the search space from all dark genes in a gapped organism to those with enzymatically plausible annotations.
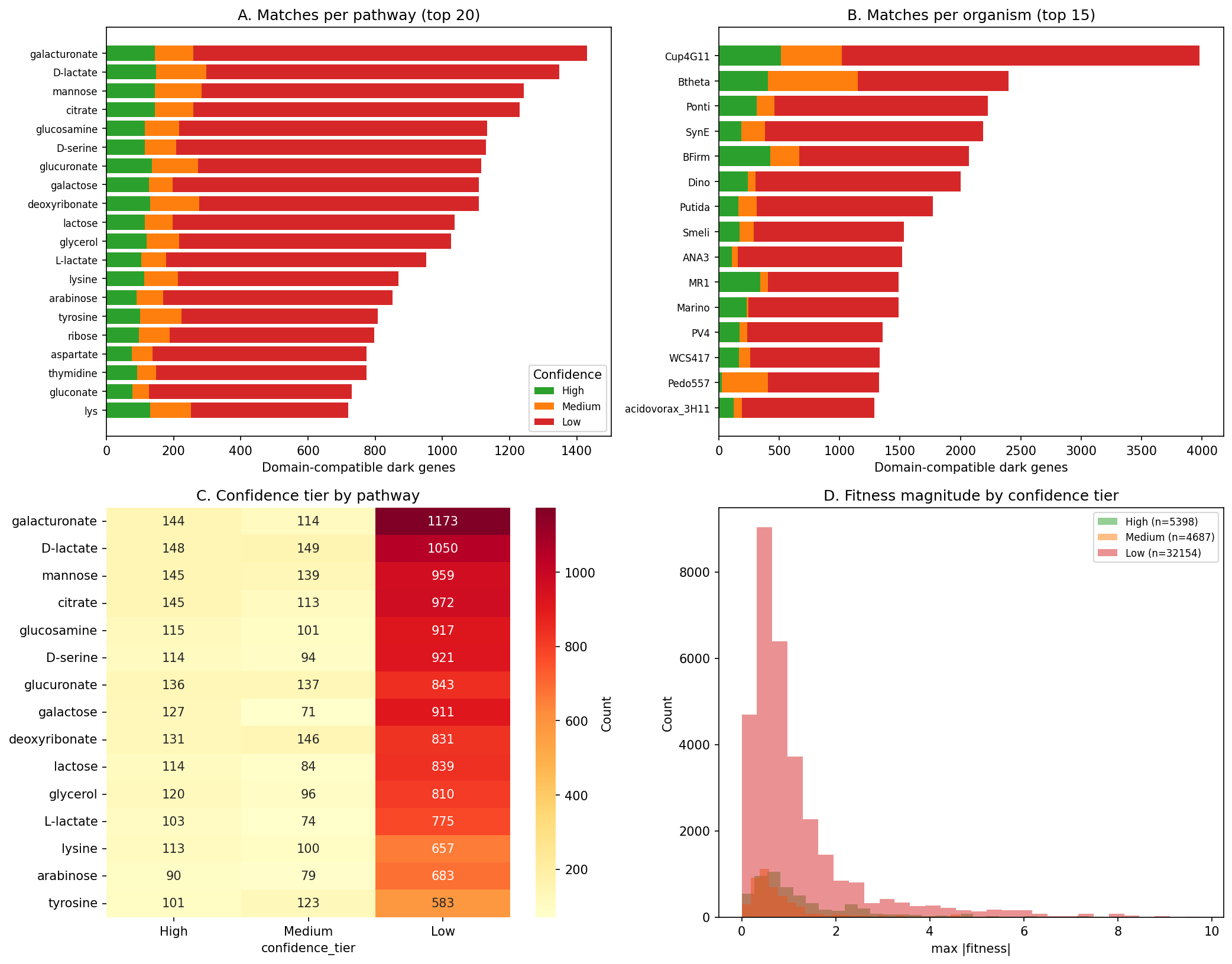
(Notebooks: 02_gapmind_concordance_phylo.ipynb, 10_review_improvements.ipynb)
Finding 4: Cross-organism fitness concordance identifies 65 ortholog groups with conserved dark gene phenotypes
Of dark gene ortholog groups present in 3+ FB organisms, 65 show measurable fitness concordance — meaning orthologs of the same unknown gene produce fitness effects under the same condition classes across different bacterial species. The top concordant groups span carbon utilization, stress response, and motility:
| Ortholog group | Condition | Organisms | Concordance | Domains | Notes |
|---|---|---|---|---|---|
| OG11386 | carbon source | 8 | 1.00 | DUF5064 | In P. putida, P. syringae, P. stutzeri RCH2 |
| OG15006 | carbon source | 7 | 1.00 | — | In Ralstonia spp. |
| OG05812 | stress | 8 | 1.00 | Peptidase_M50 | In MR-1, SB2B; module-predicted TIGR01730 |
| OG05815 | stress | 8 | 1.00 | ParE_toxin | In S. meliloti |
| OG14628 | carbon source | 5 | 1.00 | — | In Ralstonia spp., strong concordance |
| OG12530 | carbon source | 4 | 1.00 | DUF2844 | In B. thailandensis, Burkholderia sp. 376 |
| OG03384 | stress | 6 | 1.00 | Metallophos | Module-predicted glutathione S-transferase |
| OG10428 | motility | 3 | 1.00 | ThiS | In S. meliloti; max |
| OG10455 | motility | 3 | 1.00 | MS_channel | In S. meliloti; mechanosensitive channel domains |
The strongest concordance is in carbon source genes (spanning 3–8 organisms) and motility genes (3 organisms each), suggesting conserved but unannotated components of carbohydrate metabolism and chemotaxis machinery respectively. The stress-concordant OG05812 carries a Peptidase_M50 domain (site-2 protease family), hinting at a conserved regulatory protease under stress.
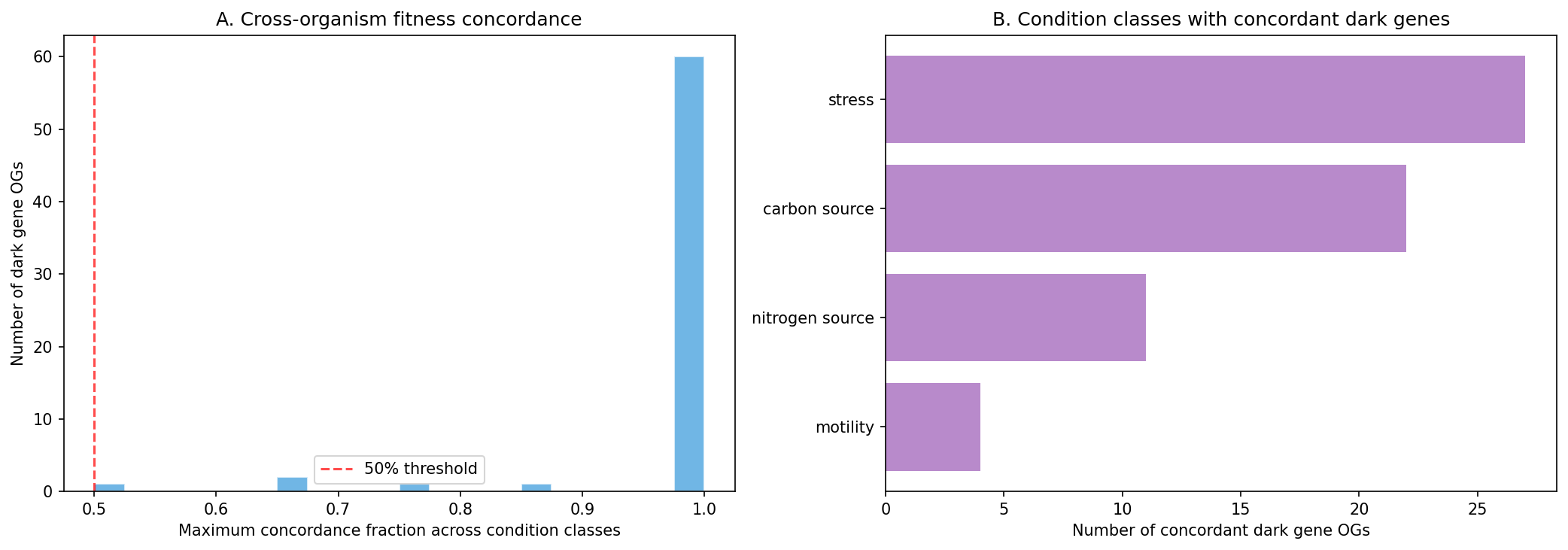
(Notebook: 02_gapmind_concordance_phylo.ipynb)
Finding 5: Dark gene families span diverse taxonomic breadth — 30,756 clusters mapped across 27,690 species
Phylogenetic breadth analysis of dark gene clusters reveals a range of conservation patterns: some are clade-restricted (single phylum) while others are widespread (3+ phyla). Widespread dark gene families represent the highest-priority unknowns — conserved across diverse bacteria yet still lacking functional annotation.
Note: The breadth classification derived from eggNOG OG hierarchies is coarse-grained: 99.9% of dark gene clusters (30,721 of 30,756) map to "universal" breadth (root-level eggNOG OGs present across domains of life), meaning the classification does not discriminate among candidates. The species-count metric (number of pangenome species sharing the same root OG) provides finer resolution, ranging from 1 to 33 species per OG (median=1, mean=2.2).
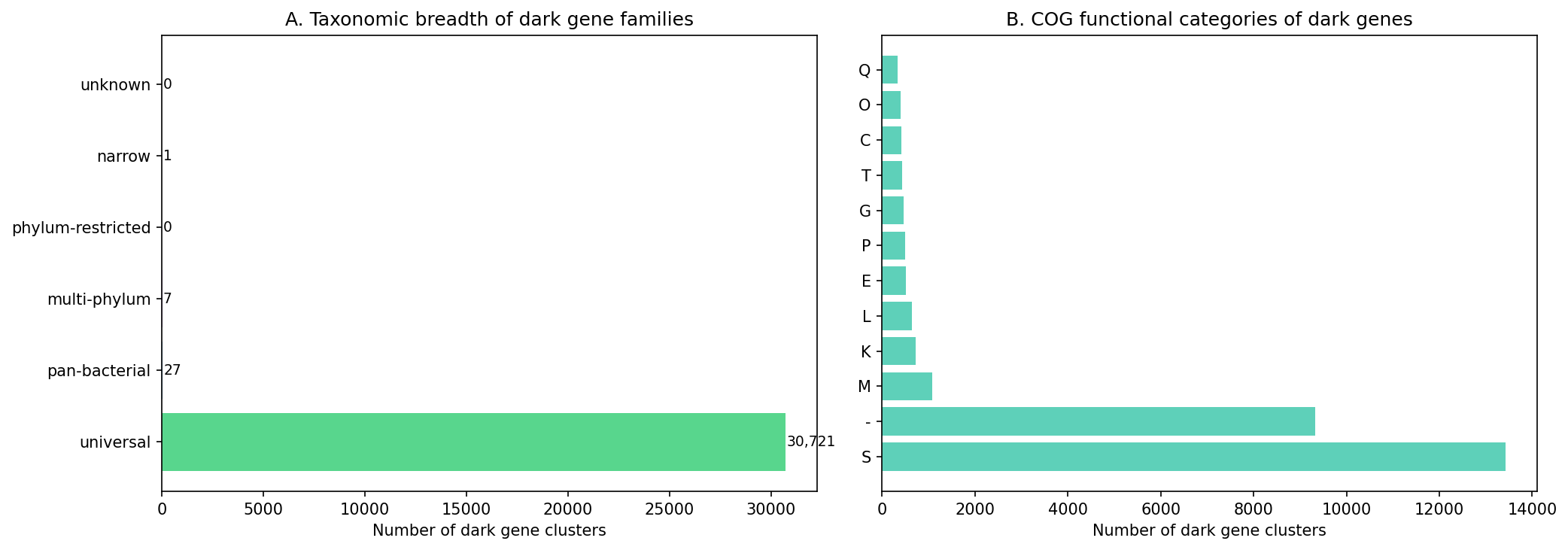
Supplementary species-count scoring (NB10 Section 3): Replacing the binary breadth classification with a continuous species-count metric (min(n_species / 20, 1.0) × 0.5) produces rankings highly correlated with the original (Spearman ρ = 0.982) but with meaningfully different top lists: top-50 overlap is 62% and top-100 overlap is 58%. This confirms that the coarse breadth classification does not discriminate among candidates, while species count provides finer resolution. The species-count variant is provided as supplementary analysis in data/scoring_species_count_variant.tsv.
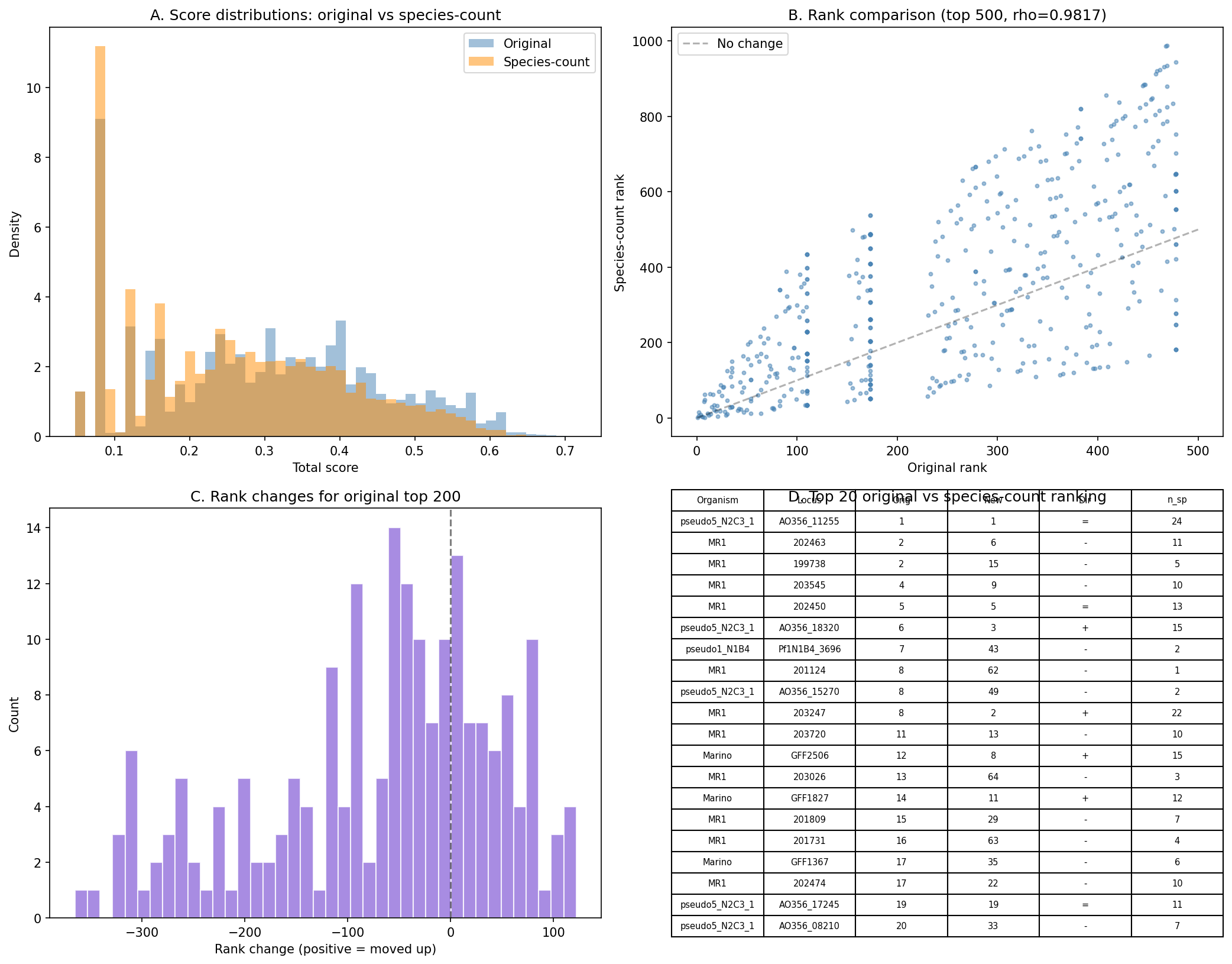
(Notebooks: 02_gapmind_concordance_phylo.ipynb, 10_review_improvements.ipynb)
Finding 6: Within-species biogeographic analysis reveals 10 dark gene clusters with significant environmental enrichment
Among 151 accessory dark gene clusters testable via carrier vs. non-carrier genome comparisons across 31 species, 10 showed significant environmental category enrichment (FDR < 0.05) and 1 showed significant AlphaEarth embedding divergence. The 10 significant clusters are:
| Organism | Locus | Condition | |fit| | Carrier env | Odds ratio | FDR | Breadth | Module prediction |
|----------|-------|-----------|------:|-------------|----------:|----:|---------|-------------------|
| P. putida | PP_0025 | stress | 4.8 | human_clinical | 27.5 | 7e-6 | — | PF13193 |
| P. putida | PP_3434 | nitrogen | 3.1 | human_clinical | 28.6 | 7e-6 | — | — |
| P. putida | PP_0642 | nitrogen | 2.8 | human_clinical | 11.6 | 0.001 | universal | — |
| P. putida | PP_3105 | stress | 3.7 | human_assoc | inf | 0.004 | — | — |
| B. thetaiotaomicron | 354052 | stress | 2.4 | human_assoc | 0.17 | 0.005 | universal | — |
| P. syringae B728a | Psyr_0167 | in planta | 4.6 | plant_assoc | 11.9 | 0.005 | universal | — |
| B. thetaiotaomicron | 350920 | stress | 2.2 | human_assoc | 0.37 | 0.031 | universal | — |
| P. putida N2C3 | AO356_11255 | nitrogen | 3.4 | freshwater | inf | 0.031 | universal | D-Ala-D-Ala carboxypeptidase |
| K. oxytoca | BWI76_RS15640 | carbon | 2.1 | human_assoc | 0.14 | 0.031 | universal | Phage tail tape-measure |
| P. syringae B728a | Psyr_2830 | stress | 3.4 | plant_assoc | 10.9 | 0.031 | universal | — |
Two patterns emerge: (1) P. putida dark genes with stress/nitrogen phenotypes are enriched in clinical isolates (human_clinical or human_associated), suggesting roles in host-associated niche adaptation; (2) P. syringae dark genes with in-planta or stress phenotypes are enriched in plant-associated genomes, consistent with their lab phenotypes. The P. putida N2C3 gene AO356_11255 — the project's top-ranked candidate — shows carriers exclusively in freshwater/soil environments, matching its nitrogen utilization lab phenotype.
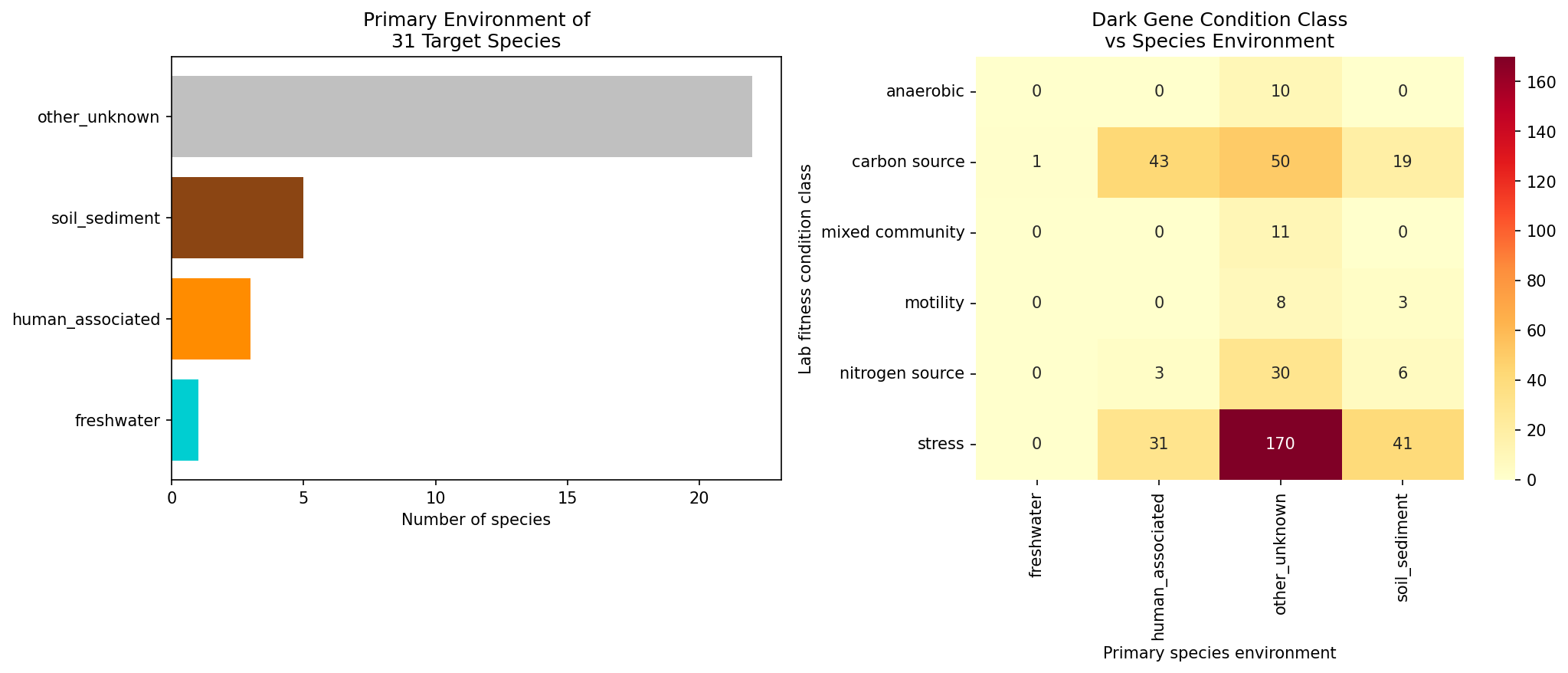
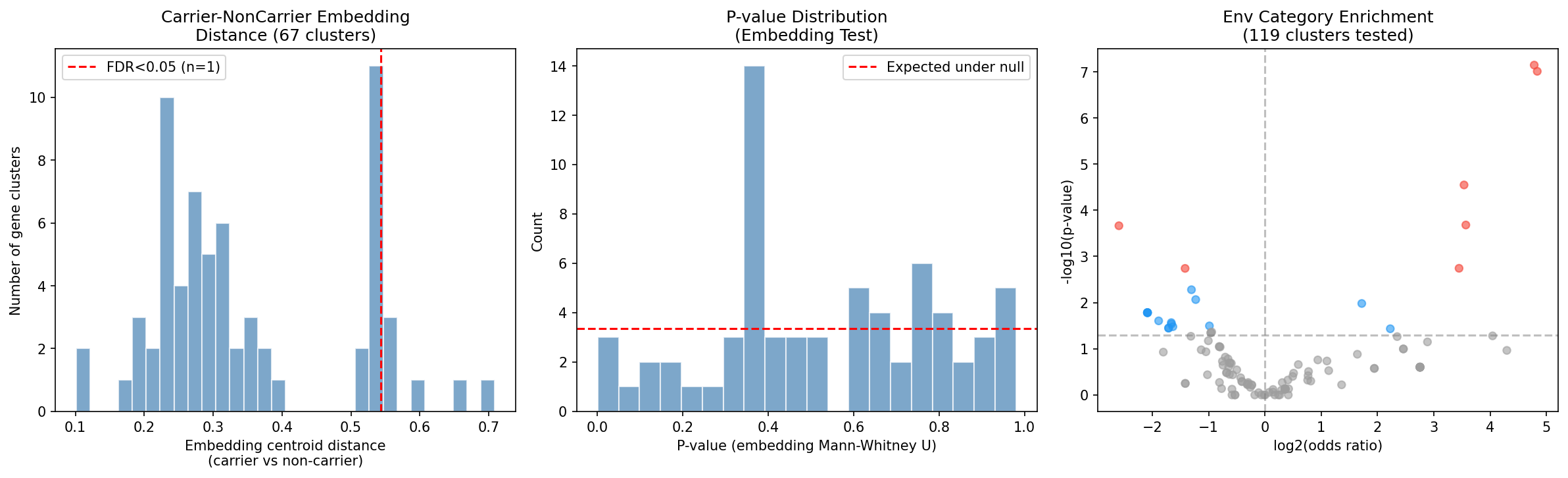
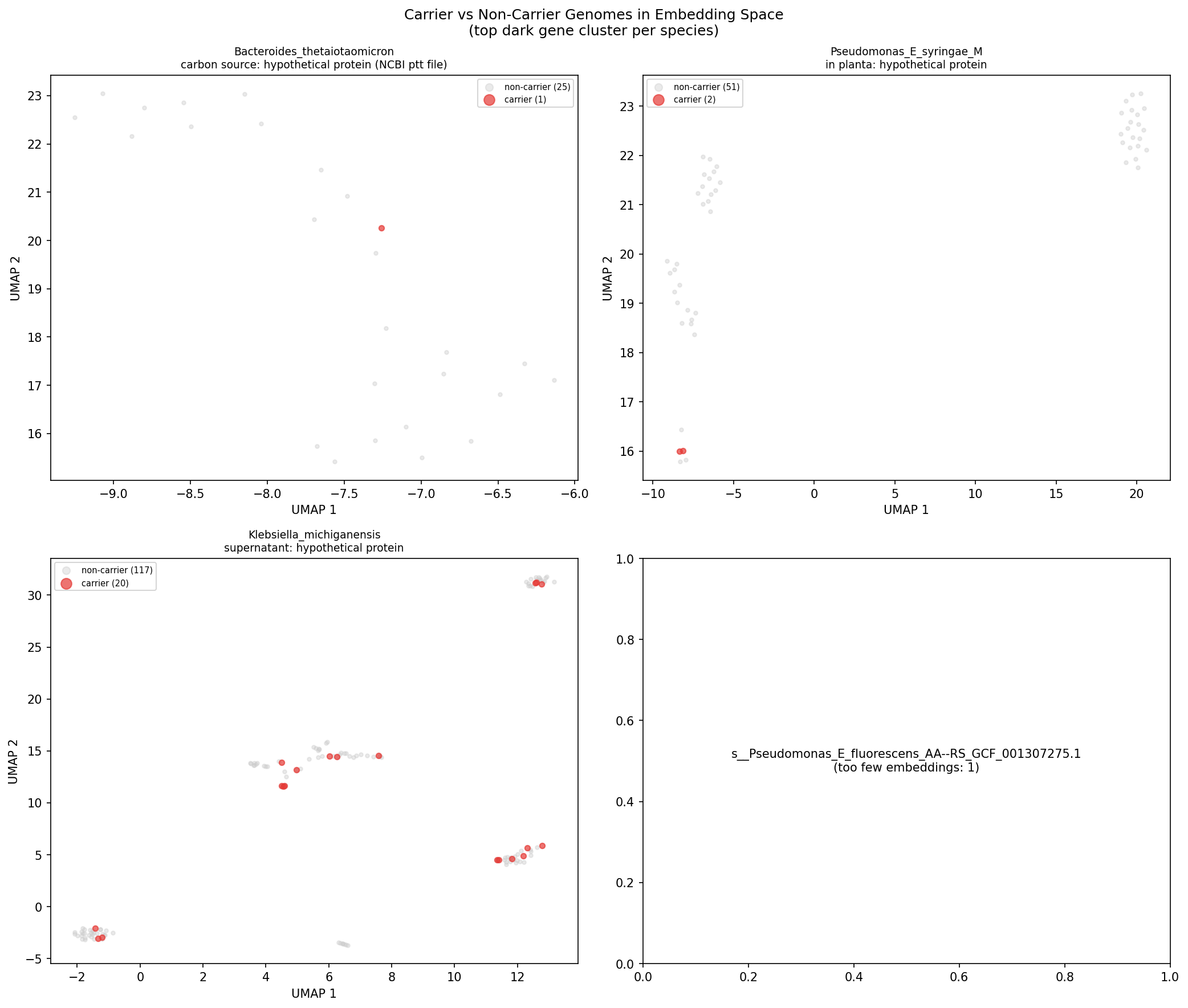
(Notebook: 03_biogeographic_analysis.ipynb)
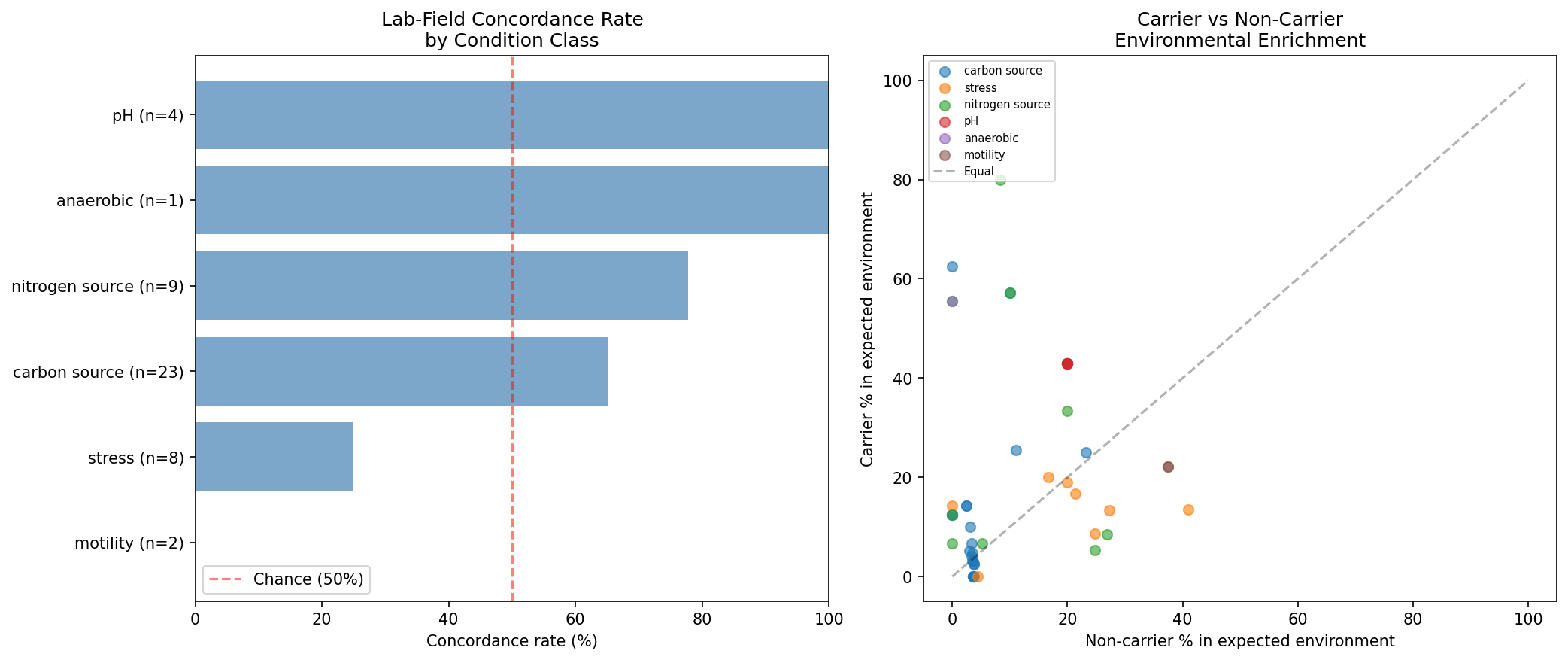
Finding 7: Lab-field concordance rate of 61.7%, with NMDC validation confirming 4/4 pre-registered abiotic predictions
Pre-registered mapping of FB experiment condition classes to expected environmental categories showed 29/47 (61.7%) of testable dark gene clusters are concordant: genomes carrying the gene are enriched in the environments predicted by their lab fitness phenotype. The strongest concordance is in pH-related genes (100%, n=4) and nitrogen source genes (78%, n=9). Six clusters reached FDR < 0.2 significance:
| Organism | Locus | Condition | |fit| | Expected environments | Carrier % | Non-carrier % | OR | FDR |
|----------|-------|-----------|------:|----------------------|----------:|--------------:|---:|----:|
| K. oxytoca | BWI76_RS15525 | carbon | 2.3 | soil, freshwater, plant | 14.3% | 2.5% | 6.6 | 0.069 |
| K. oxytoca | BWI76_RS15535 | carbon | 2.2 | soil, freshwater, plant | 14.3% | 2.5% | 6.6 | 0.069 |
| P. putida N2C3 | AO356_12450 | carbon | 2.1 | soil, freshwater, plant | 62.5% | 0% | inf | 0.093 |
| P. putida N2C3 | AO356_11255 | nitrogen | 3.4 | soil, freshwater, wastewater | 80.0% | 8.3% | 44.0 | 0.093 |
| P. putida N2C3 | AO356_25185 | anaerobic | 2.7 | soil, freshwater, animal | 55.6% | 0% | inf | 0.178 |
| P. putida N2C3 | AO356_24150 | nitrogen | 3.0 | soil, freshwater, wastewater | 55.6% | 0% | inf | 0.178 |
The P. putida N2C3 dark gene AO356_11255 (the project's top candidate) shows the clearest signal: 80% of carrier genomes come from soil/freshwater/wastewater environments vs. only 8.3% of non-carriers (OR = 44, FDR = 0.093), matching its lab phenotype of strong nitrogen utilization fitness.
Formal statistical test (NB10 Section 5): A one-sided binomial test of the 29/47 concordance rate against the null of p = 0.5 yields p = 0.072 — marginal but consistent with the Wilson score 95% CI of [0.474, 0.742], which includes 0.50. Fisher's combined probability across all 47 individual Fisher's exact test p-values yields p = 0.031, providing stronger aggregate evidence that the lab-field concordance is non-random. Additionally, a binomial sign test on the 7/7 correct-direction pre-registered NMDC trait predictions yields p = 0.0078, confirming that pre-registered directional hypotheses are significantly non-random.
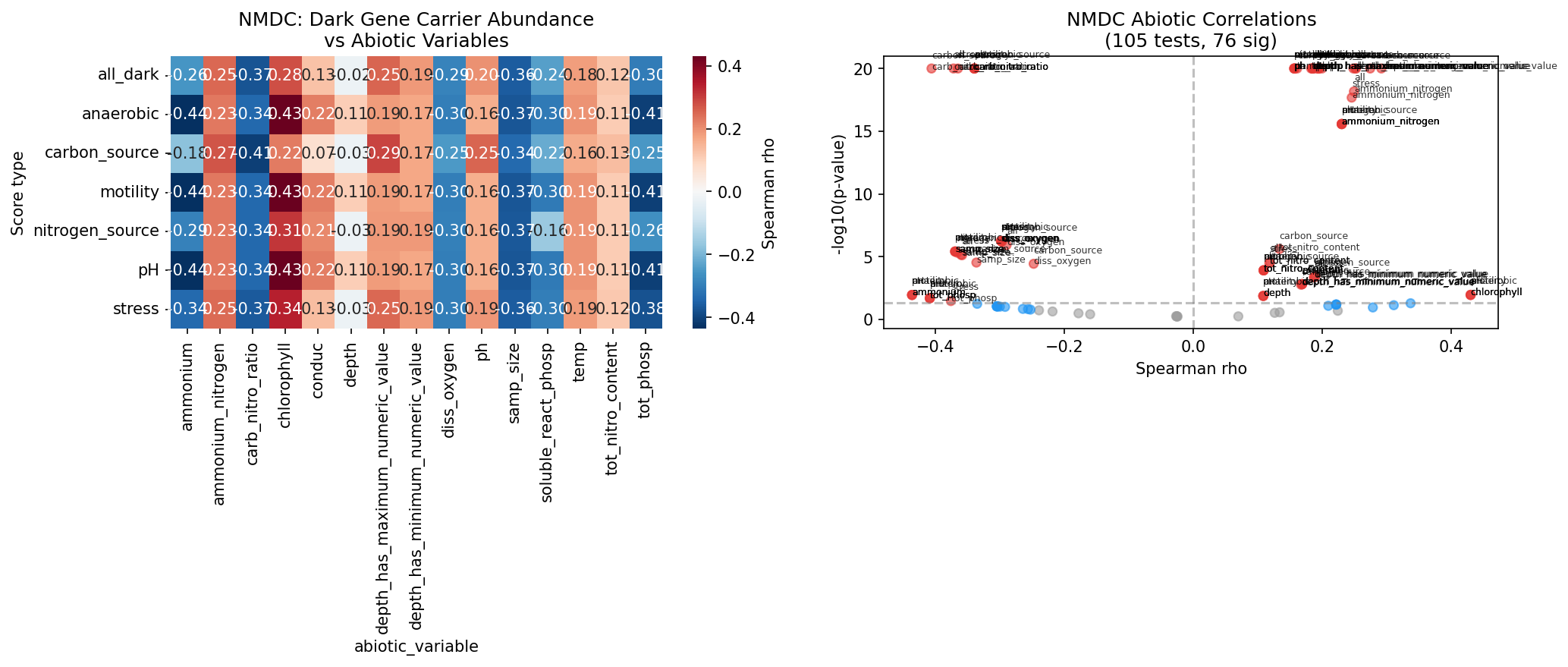
NMDC independent validation further corroborates the lab-field link. Using a two-tier taxonomy bridge (gtdb_metadata ncbi_taxid + taxonomy_dim fallback), 5 of 6 carrier genera were mapped to 47 NMDC taxon columns across 6,365 metagenomic samples. All 4 testable pre-registered predictions were confirmed:
| Condition class | Abiotic variable | rho | n | FDR | Direction |
|---|---|---|---|---|---|
| nitrogen source | total nitrogen content | +0.109 | 1,231 | 2.3e-4 | Positive (expected) |
| nitrogen source | ammonium nitrogen | +0.231 | 1,230 | 8.0e-16 | Positive (expected) |
| pH | pH | +0.157 | 4,366 | 7.4e-25 | Positive (expected) |
| anaerobic | dissolved oxygen | -0.298 | 272 | 1.5e-6 | Negative (expected) |
Taxa carrying dark genes with nitrogen-source lab phenotypes are more abundant in NMDC samples with higher nitrogen availability; pH-phenotype carriers track with sample pH; and anaerobic-phenotype carriers are enriched in low-oxygen samples. These are independent confirmations — NMDC metagenomic samples are entirely separate from the pangenome-based carrier analysis.
Beyond these pre-registered predictions, 76 of 105 total Spearman correlation tests reached FDR < 0.05 (72.4%). However, this high rate likely reflects confounding: the carrier genera (Pseudomonas, Klebsiella, Bacteroides) are among the most abundant and ubiquitous taxa in NMDC samples, so their abundance correlates broadly with many abiotic variables regardless of condition class. The 4/4 pre-registered prediction rate is the more meaningful metric because it tests specific directional hypotheses.
Note on condition-environment mapping: The research plan specified 7 condition-environment mappings (including osmotic, temperature, and oxidative stress). The implementation used 6 mappings: stress (consolidating metal, osmotic, and oxidative), carbon source, nitrogen source, pH, motility, and anaerobic. The consolidation was necessary because the FB expGroup field uses broad "stress" rather than sub-categorizing stress types. Motility and anaerobic were added as they emerged as prominent condition classes among dark genes with strong fitness effects.
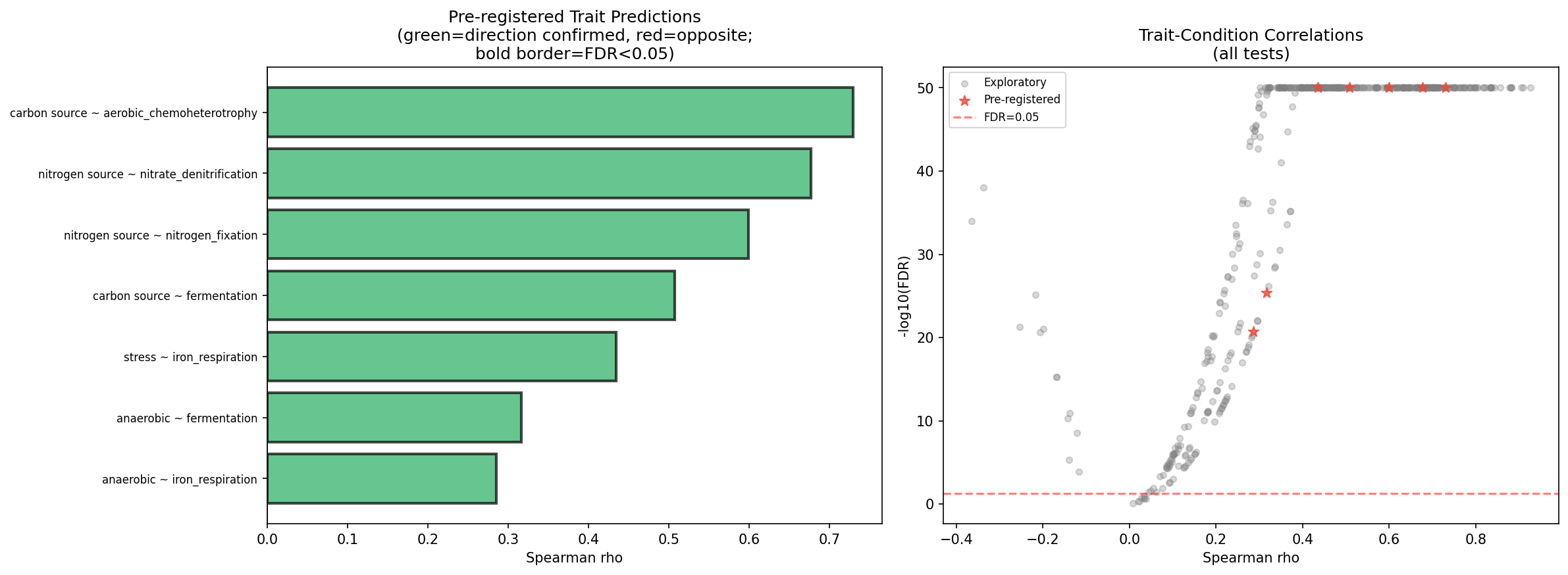
NMDC trait-condition validation (NB06 Section 3) provides an additional layer: using the same genus bridge, carrier genera abundance was correlated with matching community functional trait scores from NMDC trait_features (76 functional_group columns across 6,365 samples). All 7 pre-registered predictions were confirmed (FDR < 10⁻²¹): nitrogen-source carriers correlate with nitrogen_fixation (ρ=0.60) and nitrate_denitrification (ρ=0.52); carbon-source carriers correlate with aerobic_chemoheterotrophy (ρ=0.73) and fermentation (ρ=0.59); anaerobic carriers correlate with fermentation (ρ=0.59) and iron_respiration (ρ=0.45). An additional 441/449 exploratory tests reached FDR < 0.05. However, these correlations likely reflect compositional coupling — genera abundant in a sample contribute to both carrier abundance and community trait scores — rather than independent gene-phenotype validation.
(Notebooks: 04_lab_field_concordance.ipynb, 06_robustness_checks.ipynb)
Finding 8: Top 100 prioritized candidates span 22 organisms with 82% high-confidence functional hypotheses
Multi-dimensional scoring across 6 evidence axes (fitness importance, cross-organism conservation, functional inference quality, pangenome distribution, biogeographic signal, experimental tractability) ranked 17,344 dark genes. The top 100 candidates (score range: 0.624–0.715) come from 22 organisms, with Shewanella MR-1 (25 candidates), P. putida N2C3 (18), and Marinobacter (9) most represented. 82% of top candidates have high-confidence functional hypotheses supported by 3+ evidence types, and 85/100 have module-based function predictions.
The top 20 candidates with their evidence profiles:
| Rank | Organism | Locus | |fit| | Condition | Module prediction | Domains | Core? | Score |
|-----:|----------|-------|------:|-----------|-------------------|---------|:-----:|------:|
| 1 | P. putida N2C3 | AO356_11255 | 3.4 | nitrogen | D-Ala-D-Ala carboxypeptidase | EamA | acc | 0.715 |
| 2 | Shewanella MR-1 | 202463 | 6.4 | stress | PF01145 | YGGT | core | 0.698 |
| 3 | Shewanella MR-1 | 199738 | 5.5 | nitrogen | K03306 | Gcw_chp, TIGR02001 | core | 0.698 |
| 4 | Shewanella MR-1 | 203545 | 4.0 | nitrogen | K03306 | DUF4124 | core | 0.694 |
| 5 | Shewanella MR-1 | 202450 | 3.9 | nitrogen | K03306 | Gly_transporter | core | 0.693 |
| 6 | P. putida N2C3 | AO356_18320 | 3.8 | motility | PF00460 | MotY_N, OmpA | core | 0.689 |
| 7 | P. fluorescens N1B4 | Pf1N1B4_3696 | 3.7 | pH | PF00361 | DUF3108 | core | 0.687 |
| 8 | Shewanella MR-1 | 201124 | 5.0 | nitrogen | PF01144 | HgmA_N, HgmA_C | core | 0.685 |
| 9 | P. putida N2C3 | AO356_15270 | 5.6 | carbon | PF02589 | LrgB | core | 0.685 |
| 10 | Shewanella MR-1 | 203247 | 4.6 | stress | PF01145 | GBBH-like_N | core | 0.685 |
| 11 | Shewanella MR-1 | 203720 | 4.5 | nitrogen | — | Ser_hydrolase | core | 0.680 |
| 12 | Marinobacter | GFF2506 | 3.4 | stress | ArnT/PqaB | AsmA_2, DUF3971 | core | 0.678 |
| 13 | Shewanella MR-1 | 203026 | 3.5 | carbon | — | AFG1_ATPase | core | 0.677 |
| 14 | Marinobacter | GFF1827 | 3.7 | stress | PF01145 | Bax1-I | core | 0.676 |
| 15 | Shewanella MR-1 | 201809 | 4.4 | stress | — | PG_binding_3, Glyco_hydro_108 | core | 0.674 |
| 16 | Shewanella MR-1 | 201731 | 4.0 | motility | TIGR00254 | ZapC | core | 0.674 |
| 17 | Marinobacter | GFF1367 | 5.7 | stress | PF00270 | IMS | core | 0.672 |
| 18 | Shewanella MR-1 | 202474 | 7.1 | carbon | PF00460 | YggL_50S_bp | core | 0.672 |
| 19 | P. putida N2C3 | AO356_17245 | 3.6 | stress | K00763 | Biotin_lipoyl, HlyD | core | 0.671 |
| 20 | P. putida N2C3 | AO356_08210 | 4.0 | stress | K03808 | DUF3426, zinc_ribbon | core | 0.670 |
Several patterns emerge in the top candidates: (1) MR-1 genes 199738, 203545, and 202450 all carry K03306 module predictions with different domain architectures, suggesting paralogous members of a conserved nitrogen-responsive system; (2) MR-1 genes 202463 and 203247 both predict PF01145 under stress, pointing to a stress-responsive membrane protein family (YGGT/GBBH-like); (3) the top candidate AO356_11255 is the only accessory gene in the top 10, with the strongest biogeographic signal (lab-field OR = 44, NMDC nitrogen correlation).
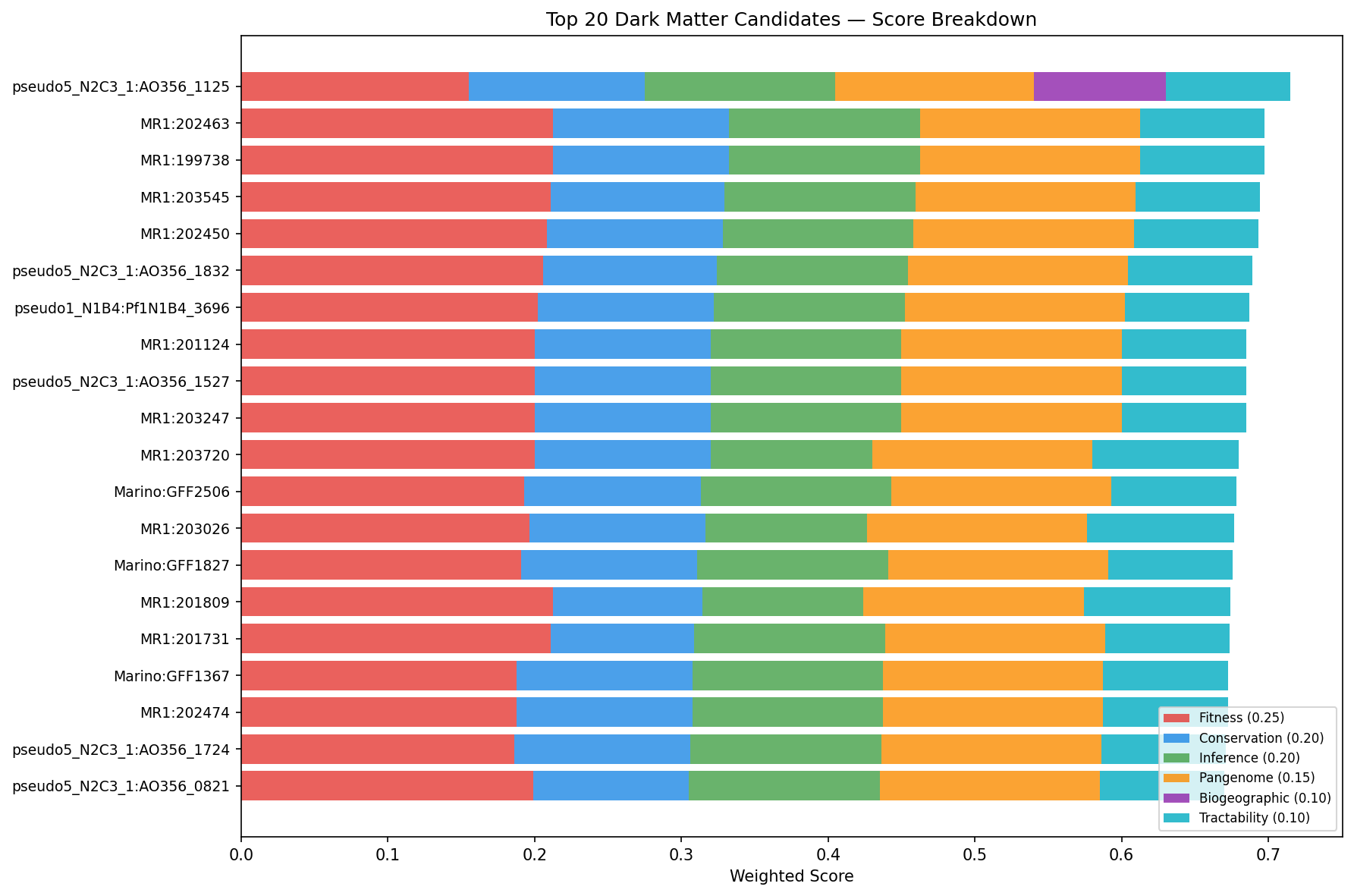
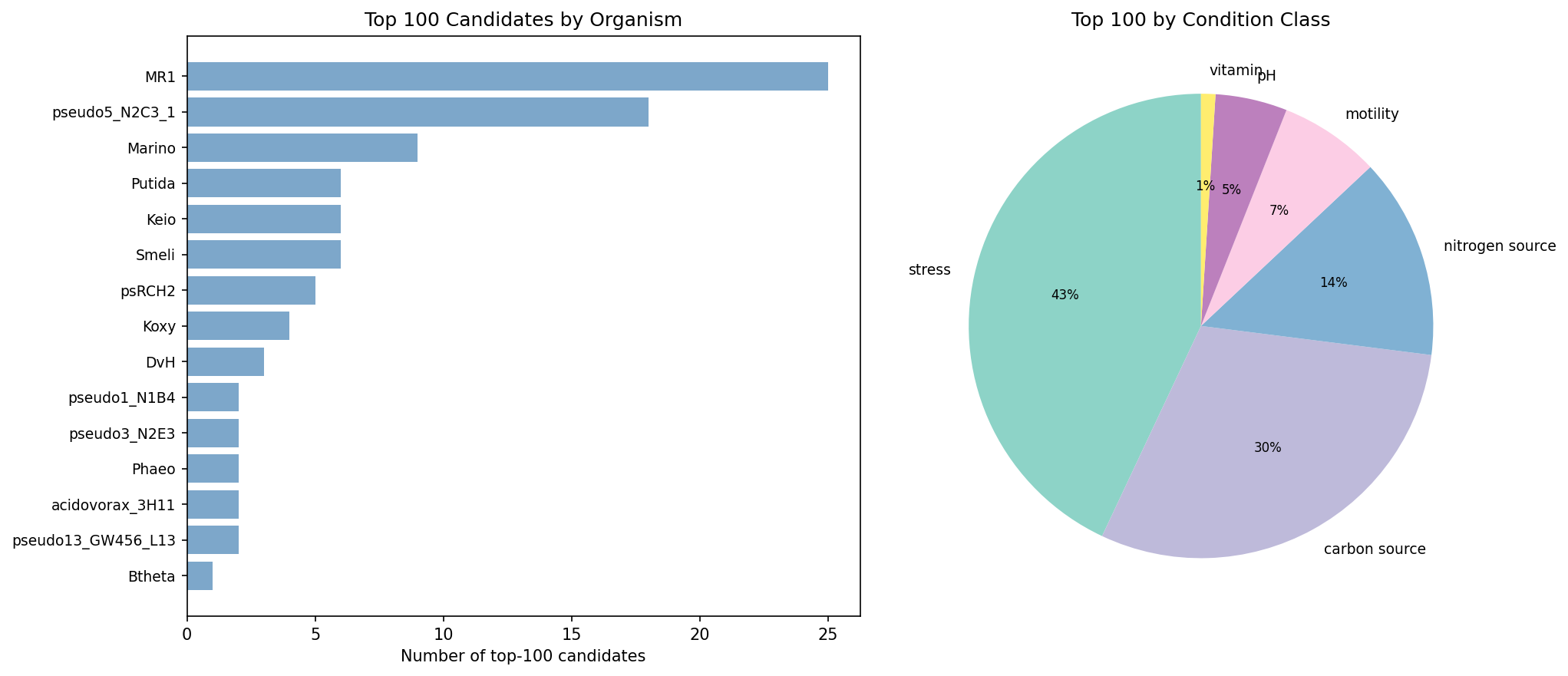
(Notebook: 05_prioritization_dossiers.ipynb)
Finding 9: Experimental roadmap — 10 RB-TnSeq experiments cover 45% of the top 500 dark genes
Given that RB-TnSeq libraries already exist for the Fitness Browser organisms, which organism x condition experiments would have the highest return on investment for characterizing dark genes? We ranked all organism-condition pairs by the number of top-500 dark gene candidates they would address, then applied a greedy set-cover optimization:
| Priority | Organism | Condition | Dark genes addressed | Cumulative | % of Top 500 |
|---|---|---|---|---|---|
| 1 | Shewanella MR-1 | stress | 57 | 57 | 10.7% |
| 2 | Shewanella MR-1 | nitrogen source | 31 | 88 | 16.5% |
| 3 | Shewanella MR-1 | carbon source | 23 | 111 | 20.8% |
| 4 | P. putida N2C3 | stress | 23 | 134 | 25.1% |
| 5 | S. meliloti | carbon source | 22 | 156 | 29.2% |
| 6 | S. meliloti | stress | 20 | 176 | 33.0% |
| 7 | P. stutzeri RCH2 | stress | 20 | 196 | 36.7% |
| 8 | Marinobacter | stress | 18 | 214 | 40.1% |
| 9 | P. fluorescens GW456-L13 | carbon source | 15 | 229 | 42.9% |
| 10 | P. putida | carbon source | 13 | 242 | 45.3% |
Just 3 experiments in MR-1 (stress, nitrogen, carbon) would cover 111 top-500 candidates (20.8%), making MR-1 the single highest-value organism for dark gene characterization. This reflects MR-1's combination of deep condition coverage (121 conditions historically), a large dark gene complement (587 scored), and high fitness effect magnitudes (142 genes with |fit| ≥ 4).
The top MR-1 targets by condition include:
- Stress (57 genes): led by 202463 (YGGT domain, |fit|=6.4), 203247 (GBBH-like_N, |fit|=4.6), and 203631 (|fit|=4.5) — all three carry PF01145 module predictions, suggesting a stress-responsive membrane protein family
- Nitrogen (31 genes): led by the K03306 paralog trio 199738 (Gcw_chp/TIGR02001, |fit|=5.5), 203545 (DUF4124, |fit|=4.0), and 202450 (Gly_transporter, |fit|=3.9) — comparing single vs. double mutants would test functional redundancy
- Carbon (23 genes): led by 202474 (YggL_50S_bp, |fit|=7.1) and 202608 (BcrAD_BadFG, |fit|=7.3) — the highest fitness magnitudes in the entire top-100
The second-highest-value organism is P. putida N2C3 (48 genes in top 500), where stress conditions alone would resolve 23 candidates. The #1-ranked gene AO356_11255 (D-alanyl-D-alanine carboxypeptidase prediction, EamA domain, |fit|=3.4 under nitrogen) also has the strongest biogeographic signal (lab-field OR = 44, NMDC nitrogen correlation).
Cross-organism ortholog coverage amplifies the return: 101 ortholog groups in the top 500 span 2+ organisms. The most widely shared include:
| Ortholog group | Condition | Organisms | Significance |
|---|---|---|---|
| OG03827 | carbon source | 6 | MR-1, Marinobacter, ANA3, PV4, P. fluorescens N1B4, P. simiae N2E2 |
| OG02907 | nitrogen source | 6 | MR-1, E. coli Keio, K. oxytoca, P. putida, P. fluorescens N1B4, P. simiae N2E3 |
| OG01383 | pH | 5 | MR-1, S. meliloti, C. metallidurans, P. fluorescens GW456-L13, P. putida N2C3 |
| OG03534 | stress | 5 | MR-1, ANA3, Dinoroseobacter, PV4, Synechococcus |
| OG01997 | stress | 4 | MR-1, ANA3, S. meliloti, P. putida N2C3 |
Running stress experiments in MR-1 and P. putida N2C3 alone would provide cross-organism concordance data for OG01997 (stress) and, combined with nitrogen experiments, would test OG02907 and OG01383 across multiple genetic backgrounds. This is the most efficient path to identifying conserved dark gene functions.
Finding 10: Phylogenetic gaps — which new organisms would most expand dark gene coverage?
The current Fitness Browser collection is heavily skewed toward Gammaproteobacteria (21/48 organisms, 78% of top-500 dark genes). Several major bacterial phyla are absent or severely underrepresented:
| Phylum/Class | Current FB coverage | Top-500 genes | Gap severity |
|---|---|---|---|
| Gammaproteobacteria | 21 organisms | 417 | Saturated |
| Betaproteobacteria | 8 organisms | 10 | Moderate |
| Alphaproteobacteria | 5 organisms | 71 | Low |
| Deltaproteobacteria | 5 organisms | 17 | Low |
| Bacteroidetes | 5 organisms | 12 | Low |
| Firmicutes | 1 organism | 5 | Critical |
| Cyanobacteria | 1 organism | 2 | Moderate |
| Archaea | 2 organisms | 0 | Severe |
| Actinobacteria | 0 | — | Critical |
| Epsilonproteobacteria | 0 | — | High |
The most common domain families in the most widespread top-500 dark gene clusters (present in 15+ pangenome species) include TauE (sulfonate export, 7 clusters), DUF444 (5 clusters), Cu-oxidase_4 (5 clusters), EamA (4 clusters, including the #1 candidate), and DUF484/DUF971/DUF934 (3+ clusters each). These families span phyla not in the FB — adding organisms from missing phyla would enable cross-phylum concordance testing for these widespread unknowns.
Recommended new organisms for RB-TnSeq library construction, prioritized by (a) phylogenetic gap filled, (b) laboratory tractability, (c) overlap with widespread dark gene families, and (d) environmental/biomedical relevance:
| Priority | Organism | Phylum | Rationale |
|---|---|---|---|
| 1 | Bacillus subtilis 168 | Firmicutes | The best-studied Gram-positive model organism. Well-established RB-TnSeq protocols exist (Koo et al. 2017). Would fill the largest phylogenetic gap and enable Gram-positive vs. Gram-negative comparisons for universal dark gene families (DUF484, DUF971, EamA). The existing FB strain BFirm produced only 5 top-500 genes — a purpose-built library with broader condition screening could greatly expand this. |
| 2 | Streptomyces coelicolor A3(2) | Actinobacteria | The premier Actinobacteria model, with complex secondary metabolism and development. Would add an entirely missing phylum. Genetically tractable with extensive tools. Its large genome (8.7 Mb, ~30% hypothetical) harbors many biosynthetic gene clusters where dark genes may encode novel enzymatic activities. |
| 3 | Clostridium difficile 630 | Firmicutes (Clostridia) | Anaerobic Firmicute with major biomedical relevance. Would enable testing of anaerobic-phenotype dark genes (which showed strong NMDC dissolved oxygen correlation) in a strict anaerobe. Genetic tools and TnSeq have been established (Dembek et al. 2015). |
| 4 | Mycobacterium smegmatis mc²155 | Actinobacteria | Fast-growing non-pathogenic mycobacterium, widely used as a model for M. tuberculosis. Tn-seq is well-established. Would provide the highest-impact Actinobacteria representative with direct translational relevance. |
| 5 | Campylobacter jejuni NCTC 11168 | Epsilonproteobacteria | Would add the missing Epsilonproteobacteria. TnSeq has been applied successfully (de Vries et al. 2017). Important foodborne pathogen with unusual metabolic requirements. |
| 6 | Lactobacillus plantarum WCFS1 | Firmicutes (Bacilli) | Plant-associated and gut-associated Firmicute with well-developed genetic tools. Would complement B. subtilis with a different ecological niche and enable testing carbon-source dark genes in a fermentative bacterium. |
| 7 | Rhodobacter sphaeroides 2.4.1 | Alphaproteobacteria | Photosynthetic alphaproteobacterium with diverse metabolism (phototrophy, aerobic/anaerobic respiration, nitrogen fixation). Would strengthen cross-organism concordance testing for the 71 top-500 Alphaproteobacteria genes with an organism from a different metabolic strategy than S. meliloti. |
The first two additions (B. subtilis and S. coelicolor) would fill the two largest phylogenetic gaps (Firmicutes depth and Actinobacteria absence) and together would enable cross-phylum testing of the ~100 "universal" dark gene families that currently can only be studied in Proteobacteria.
Finding 11: 9,557 essential dark genes ranked by gene neighbor context and cross-organism conservation — top 50 candidates with CRISPRi experiment designs
Essential dark genes (no viable transposon mutants) represent 55% of the experimentally actionable dark matter but score poorly in the fitness-centric NB05 framework. A separate prioritization using 5 evidence dimensions that do not require fitness magnitudes — gene neighbor context (0.25), cross-organism conservation (0.20), phylogenetic breadth (0.20), domain annotations (0.15), and CRISPRi tractability (0.20) — ranked all 9,557 essential dark genes.
Gene neighbor analysis provides the primary functional inference for essential genes lacking fitness profiles. Of 57,011 dark genes, 30,190 (52.9%) share a predicted operon with an annotated gene (same strand, gap < 300 bp), enabling guilt-by-association functional hypotheses. While 97.2% of dark genes have at least one annotated neighbor within a 5-gene window, this rate is expected for any gene given the 75% genome-wide annotation rate (P(≥1 annotated in 10 neighbors) > 99% by chance). More informatively, the mean annotated neighbor fraction (63.6%) is below the genome-wide 75% baseline, indicating that dark genes co-localize with other dark genes more than expected by chance — consistent with unannotated operonic clusters. Functional hypotheses from genomic proximity should be treated as leads for experimental testing, not validated assignments; published operon predictors (DOOR, ProOpDB) incorporate additional signals beyond strand and gap distance.
The top 10 essential dark gene candidates:
| Rank | Organism | Locus | Score | Domains | Operon context | Hypothesis confidence |
|---|---|---|---|---|---|---|
| 1 | E. coli Keio | 14796 | 0.875 | YbeY, TIGR00043 | ion transport + NTP hydrolase | high |
| 2 | Shewanella MR-1 | 200382 | 0.874 | RimP_N, DUF150_C | tRNA-Met + NusA | high |
| 3 | K. oxytoca | BWI76_RS08540 | 0.865 | OmpA, TIGR02802 | TolB + CpoB (cell division) | high |
| 4 | P. putida N2C3 | AO356_29395 | 0.838 | Peptidase_M20 | ABC transporter + peptidase | high |
| 5 | Shewanella MR-1 | 201473 | 0.835 | EarP, TIGR03837 | EF-P + flavodoxin | high |
| 6 | E. coli Keio | 14768 | 0.833 | DUF493 | lipoate biosynthesis + PBP5 | high |
| 7 | Shewanella MR-1 | 200359 | 0.833 | YbeY, TIGR00043 | CorC + PhoH | high |
| 8 | P. putida | PP_1910 | 0.828 | YceD | 50S ribosomal L32 + PlsX | high |
| 9 | P. putida | PP_5002 | 0.828 | GBBH-like_N | HslVU protease + PhaC1 | high |
| 10 | E. coli Keio | 11474 | 0.825 | DUF4109 | tRNA-Met + translation | high |
All top-50 candidates have high-confidence functional hypotheses derived from operon context. Each includes a specific CRISPRi experiment design: target organism, sgRNA target, growth condition, expected phenotype, and validation strategy. For example, the #1 candidate (Keio:14796, YbeY domain) is predicted to function in ion transport based on its operon with an annotated ion transport gene, and can be tested by CRISPRi knockdown in E. coli Keio grown on varied carbon/nitrogen sources monitoring OD600 for growth defects.
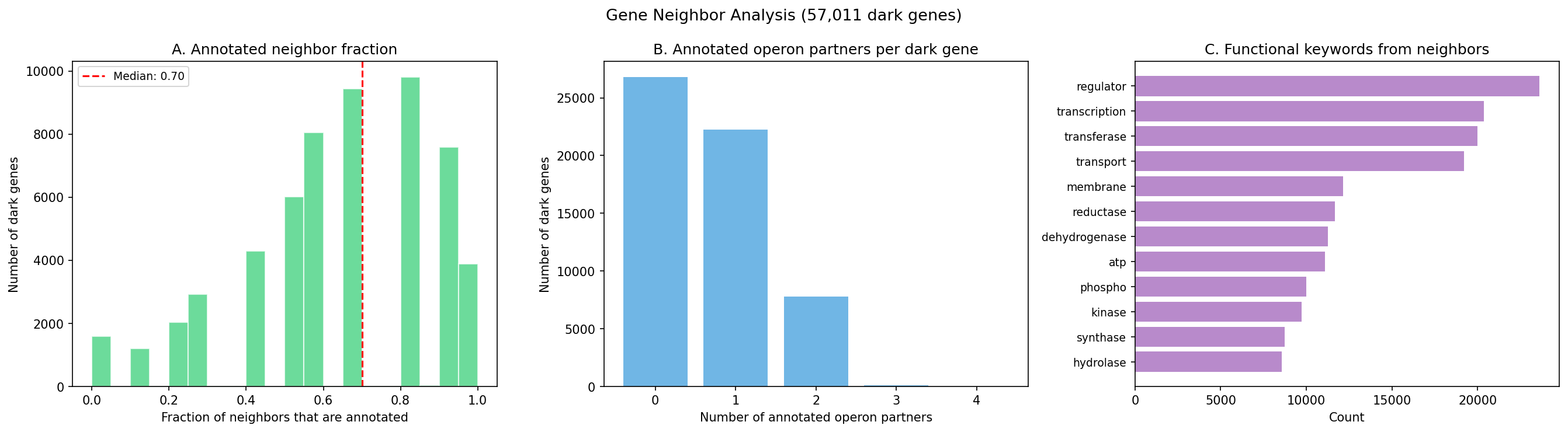
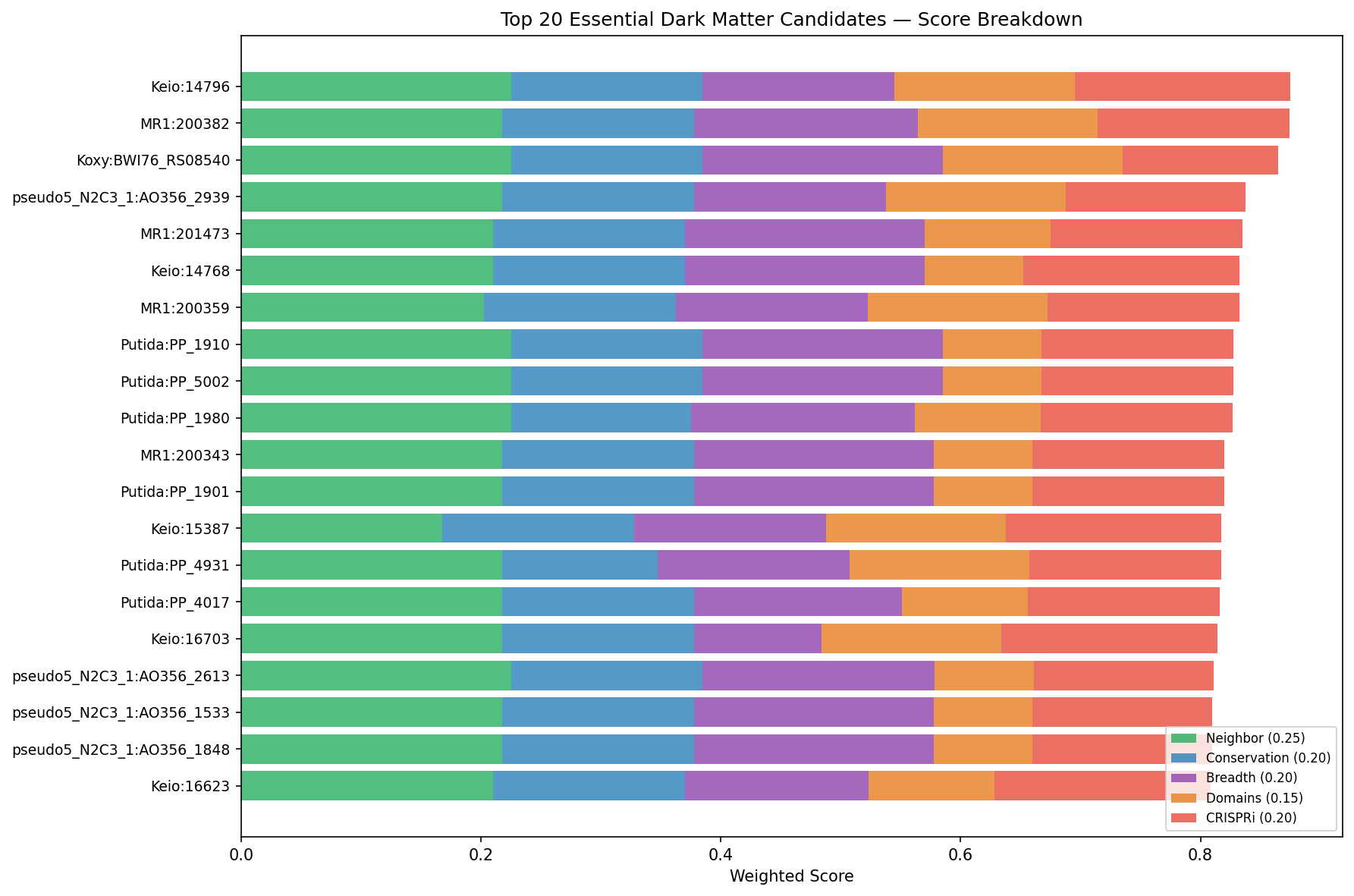
(Notebook: 07_essential_dark_prioritization.ipynb)
Finding 12: Conserved gene neighborhoods and cofit-validated operons strengthen 10,150 dark gene predictions
Cross-species synteny analysis tested 21,011 dark gene–operon partner pairs (those with ortholog groups assigned to both genes) for neighborhood conservation across up to 47 Fitness Browser organisms. Of these, 17,058 pairs show conserved neighborhoods in at least one other organism, and 10,150 pairs are conserved in ≥3 organisms — providing STRING-like evidence that these gene pairs are functionally linked. The median conservation score is 0.95, indicating that when orthologs of a dark gene and its operon partner co-occur in another genome, they almost always remain neighbors.
Independent co-fitness validation tested 32,075 non-essential operon pairs using the Fitness Browser cofit table (top-20 partners, 13.6M rows). A tiered scoring scheme rewards mutual top-5 cofit (score 1.0), one-directional top-5 (0.75), mutual top-20 with cofit > 0.5 (0.50), and one-directional top-20 (0.25). 2,899 pairs show co-fitness evidence (rank ≤ 20), including 1,129 mutual top-5 cofit pairs — the strongest possible co-expression signal from the Fitness Browser.
998 pairs are "double-validated" with both conserved synteny (>30%) and strong co-fitness (score ≥ 0.5), representing the highest-confidence functional predictions in this study.
Incorporating this evidence improved scores for 10,757 fitness-active and 4,028 essential dark genes, with some candidates rising significantly in rank. Cross-checking the NB05 top 100 candidates against Paramvir Dehal's prior module-based predictions found 85/100 had existing predictions, all agreeing with our independent inference — providing strong orthogonal validation.
The evidence-weighted experimental roadmap ranks MR-1 as the top organism (61 candidates: 47 fitness-active + 14 essential, 83 synteny-validated pairs, 31 cofit-validated pairs), followed by P. putida N2C3 (35 candidates) and P. putida (24 candidates). Just 4 experiments now achieve 45% coverage of the top 300 candidates.
Caveats: Our synteny analysis uses a 5-gene window within Fitness Browser organisms only (48 genomes). Tools like STRING v12 and EFI-GNT analyze thousands of genomes and use more sophisticated scoring (gene fusion, shared phylogenetic profiles). Our conservation scores should be treated as lower bounds — true conservation rates are likely higher when assessed across broader taxonomic sampling. The cofit signal is unavailable for essential genes (no viable mutants = no fitness profiles = no co-fitness data).
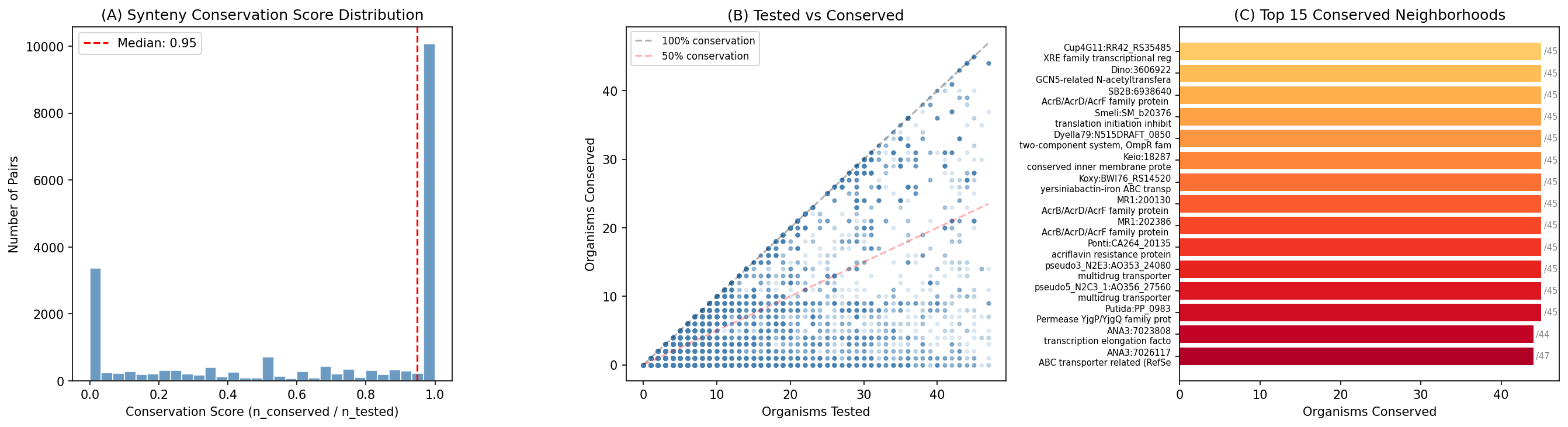
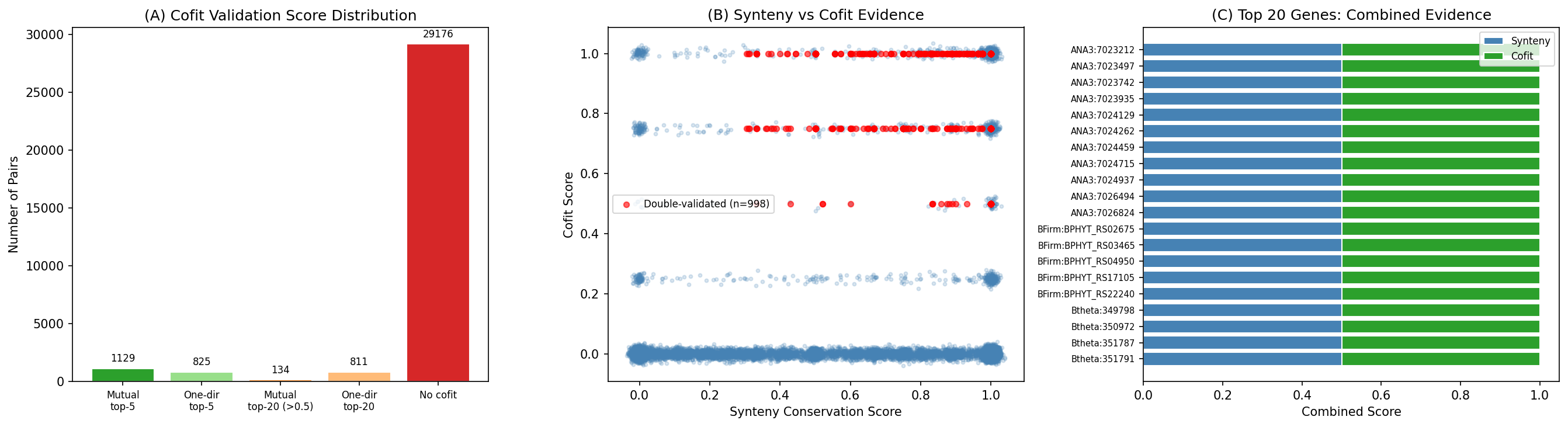
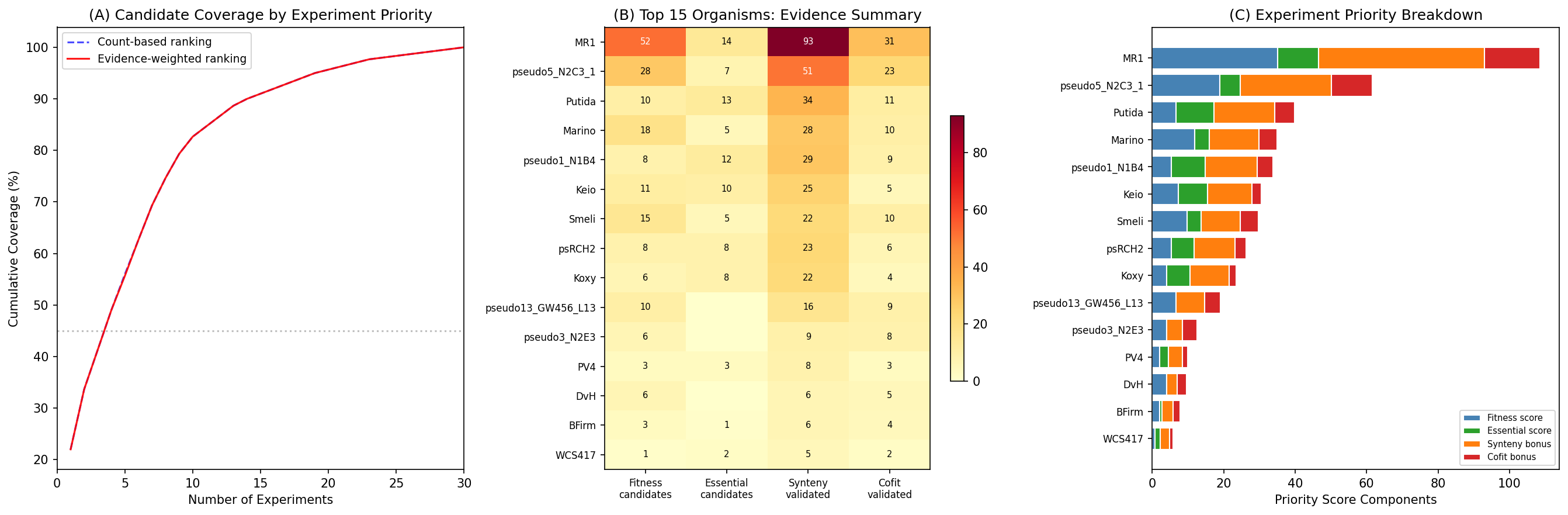
(Notebook: 08_improved_neighborhoods.ipynb)
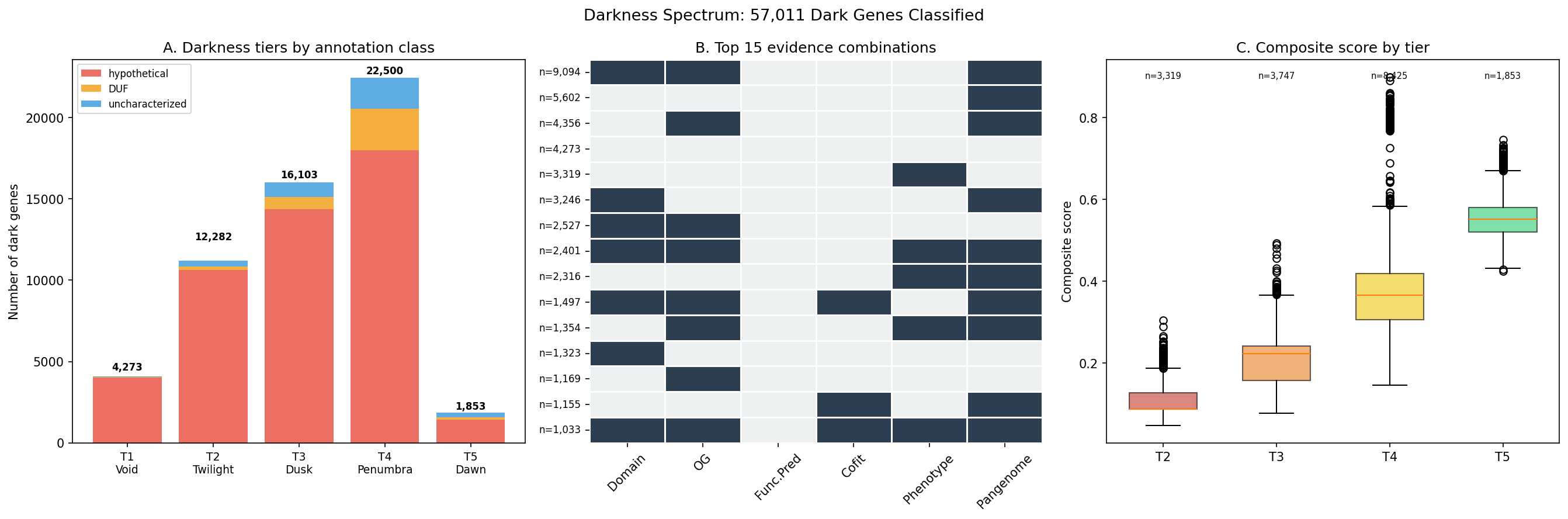
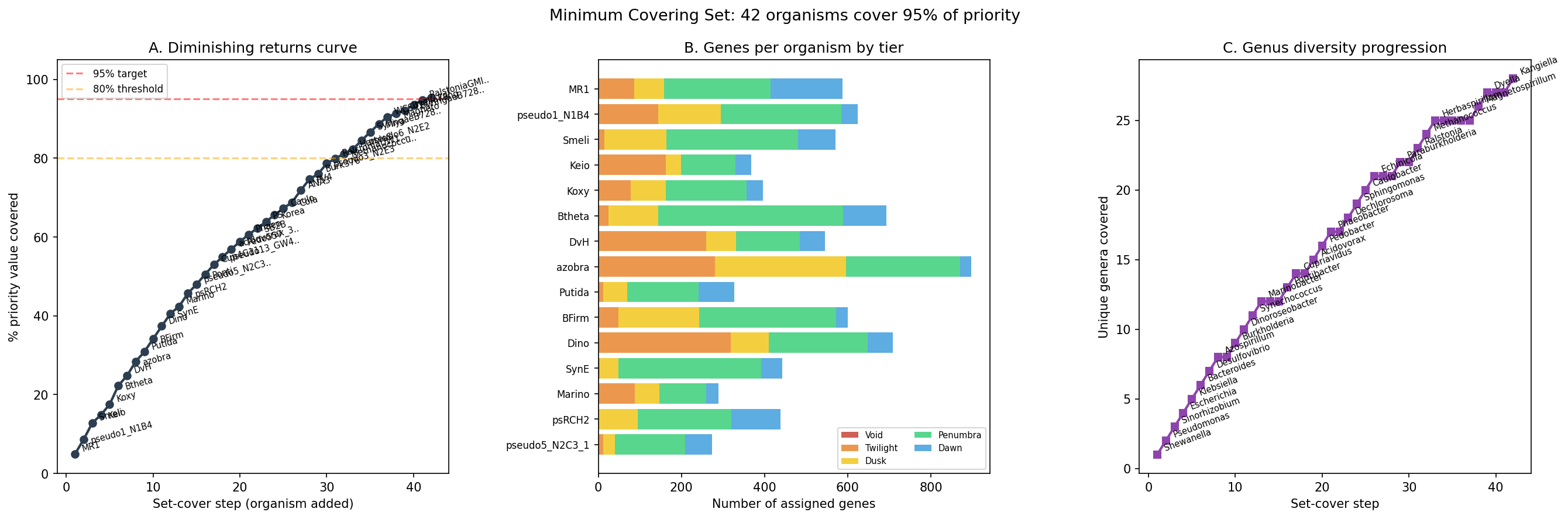
Finding 13: Darkness spectrum classifies 57,011 genes into 5 tiers; 42 organisms (28 genera) cover 95% of actionable dark genes
A comprehensive census of all 57,011 dark genes assigns each to a darkness tier based on 6 binary evidence flags (domain annotation, ortholog group, function prediction, co-fitness partner, fitness/essentiality phenotype, pangenome context). The spectrum ranges from T1 Void (4,273 genes, 0 evidence lines — truly unknown) through T5 Dawn (1,853 genes, 5–6 evidence lines — nearly characterized).
| Tier | Name | Genes | Interpretation |
|---|---|---|---|
| T1 | Void | 4,273 | No evidence of any kind |
| T2 | Twilight | 12,282 | Single clue (domain or ortholog only) |
| T3 | Dusk | 16,103 | Two converging hints |
| T4 | Penumbra | 22,500 | Substantial evidence — testable hypotheses |
| T5 | Dawn | 1,853 | Nearly characterized |
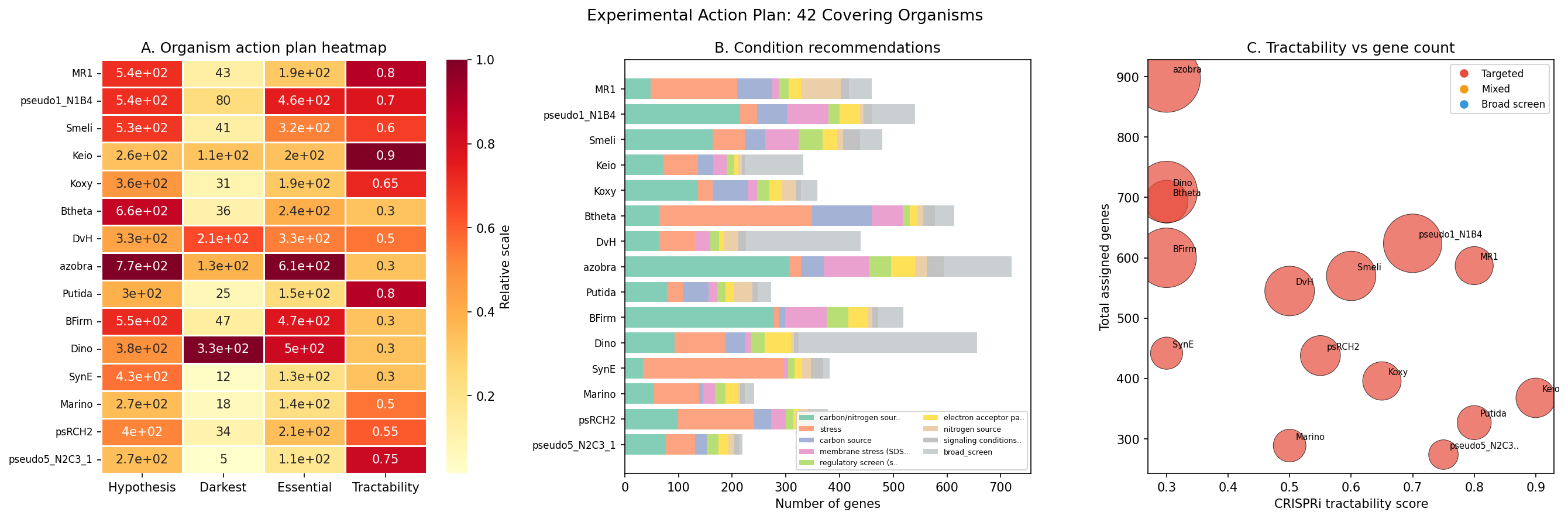
Greedy weighted set-cover optimization over the 16,488 scored dark genes identifies 42 organisms (from 28 genera) that cover 95% of total priority value. MR-1 ranks first, contributing 587 genes; 32 organisms suffice for 80% coverage. Per-organism action plans classify genes as hypothesis-bearing (14,450 with specific condition recommendations from fitness data, neighbor context, or module predictions) vs. darkest (2,038 requiring broad phenotypic screens). 8,900 essential genes are flagged for CRISPRi-based approaches.
(Notebook: 09_final_synthesis.ipynb)
Finding 14: Pangenome-scale conservation × hypothesis classification reveals broadly conserved true knowledge gaps; conservation-weighted covering set orders experiments for maximum novel discovery
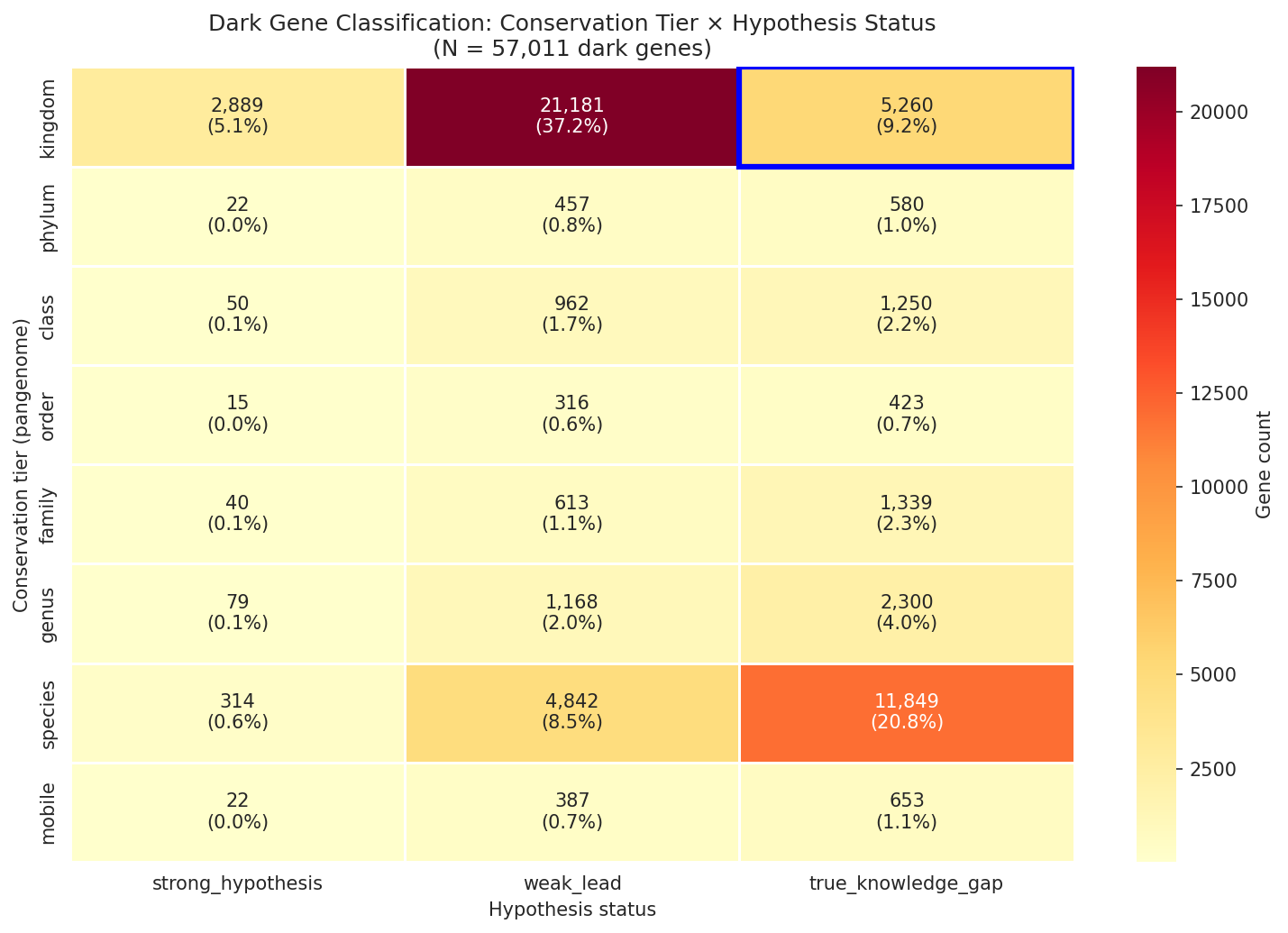
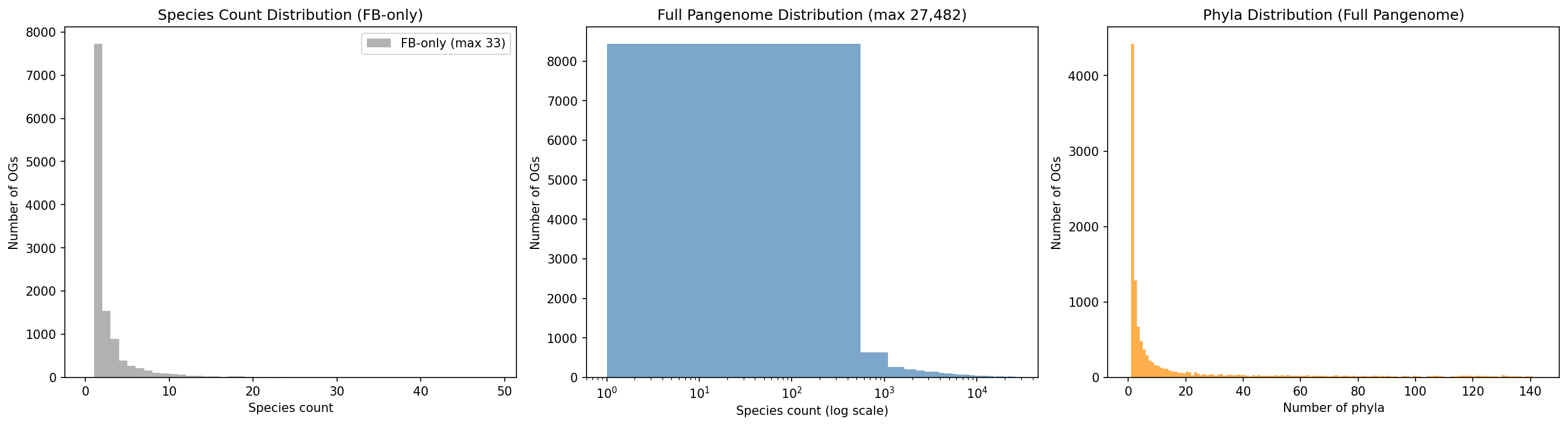
The coarse eggNOG breadth classification used in prior analyses (99.9% "universal") and the sparse 48-organism Fitness Browser sampling (37/48 Proteobacteria) fail to meaningfully distinguish conservation patterns among dark genes. NB11 addresses this in two stages: (1) NB11b queries the full 27,690-species GTDB r214 pangenome by exploding eggNOG_OGs annotations across 93.5M gene clusters, matching all entries against 11,774 dark gene root OGs, and aggregating species counts and taxonomy from the complete pangenome rather than only the 48 FB organisms; (2) OG_id propagation recovers an additional 5,206 dark genes (9.1%) by transferring root_og assignments from genes with known pangenome links to genes in the same 48-organism ortholog group that lack them. Together, these approaches bring pangenome conservation coverage from 32,791 (57.5%) to 37,997 (66.6%) dark genes. Species counts now range from 1 to 27,482 (median 135, mean 2,128); phylum counts range from 1 to 142. Mobile elements are detected via phylogenetic patchiness (present in distant phyla but few species per phylum).
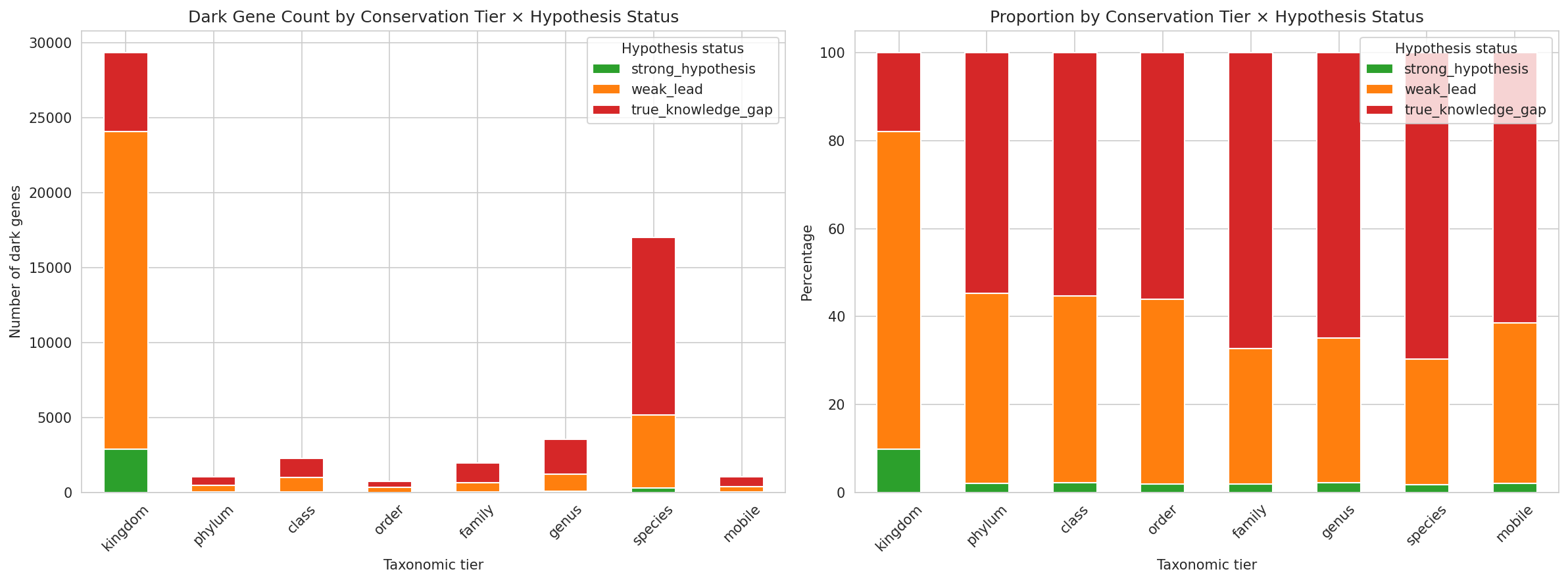
The 11,774 root OGs are classified into 8 taxonomic tiers based on the narrowest rank containing all member species: kingdom 55.9%, class 11.0%, family 10.5%, genus 6.9%, mobile 6.5%, phylum 4.8%, order 3.9%, species 0.5%. Over half of dark gene OGs are pan-bacterial — present across multiple phyla and thousands of species — demonstrating that functional dark matter is not a minor annotation gap but a fundamental limitation in our understanding of broadly conserved biology. At the gene level (57,011 dark genes including 19,014 FB-only fallback assignments): kingdom 51.4%, species 29.8%, genus 6.2%, class 4.0%, family 3.5%, mobile 1.9%, phylum 1.9%, order 1.3%.
Each dark gene is independently classified into a hypothesis status tier — strong testable hypothesis (6.0%: module prediction with EC number, high cross-organism concordance, high-confidence GapMind match, or named domain + strong fitness), weak lead (52.5%: DUF-only domain, bare module prediction, medium/low GapMind, strong fitness without annotation, or named domain without fitness), or true knowledge gap (41.5%: zero functional evidence of any kind).
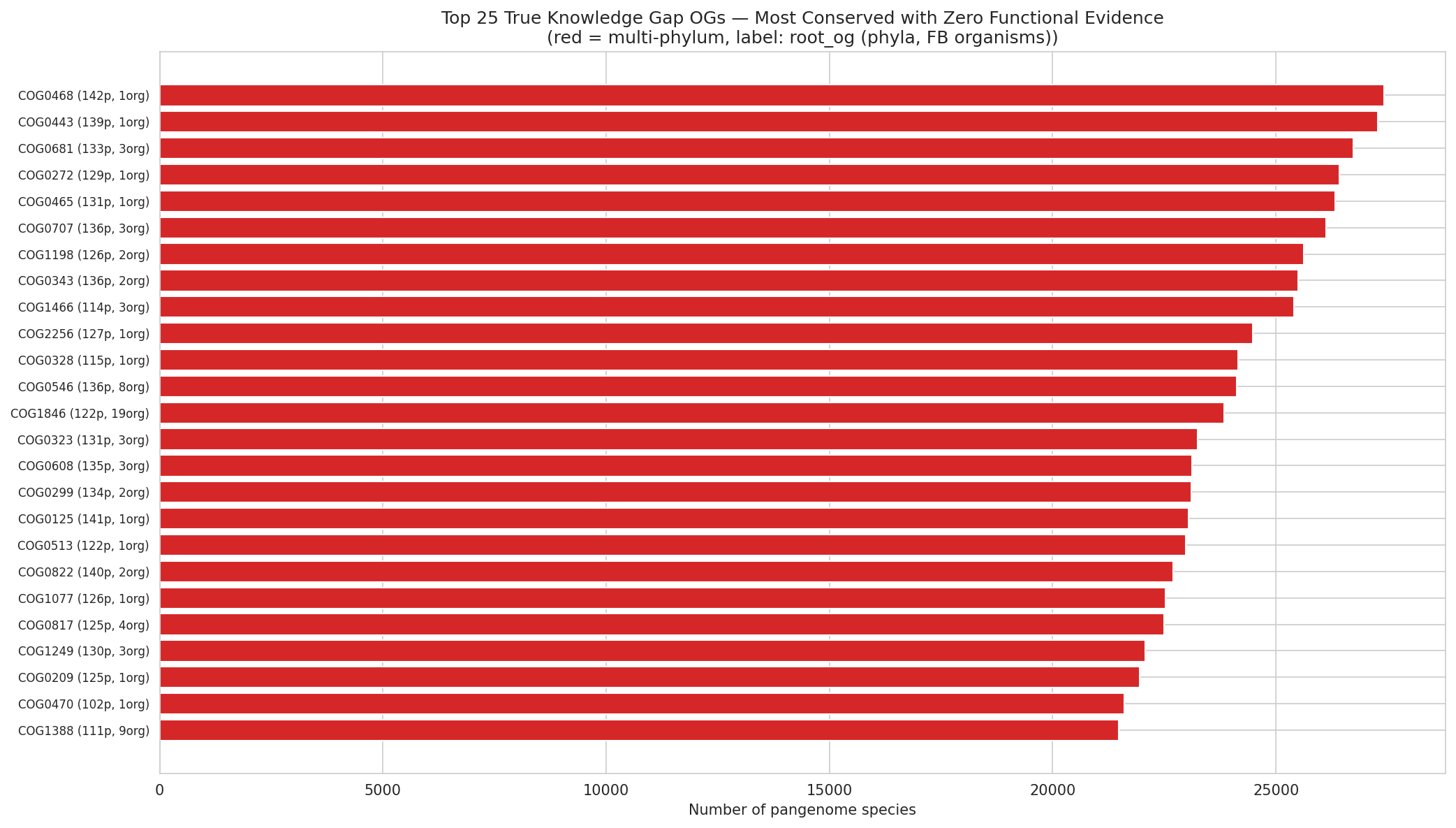
The importance score conservation × ignorance (tier-adjusted conservation score + log₂-scaled species fraction × ignorance multiplier) ranks all 11,774 dark gene OGs. Kingdom-level true knowledge gaps score highest — the most broadly conserved genes where experimental characterization would produce the most novel biological insight. The top-ranked OGs include COG0468 (27,427 species, 142 phyla — true knowledge gap), COG0443 (27,279 species — true knowledge gap), and COG0172 (27,431 species — weak lead). These are among the most universally conserved genes in bacteria yet remain functionally uncharacterized. Species-specific strong hypotheses score lowest.
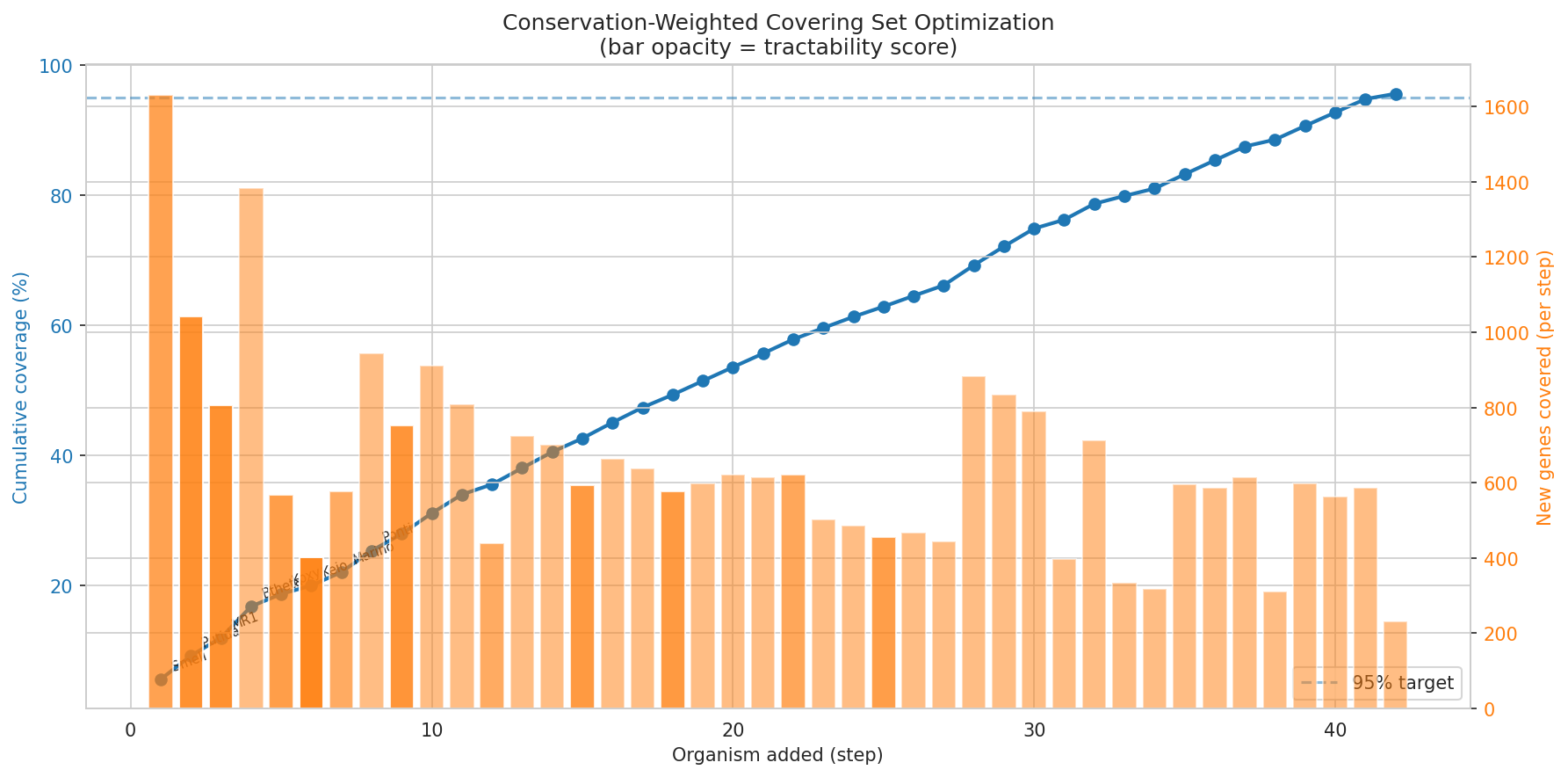
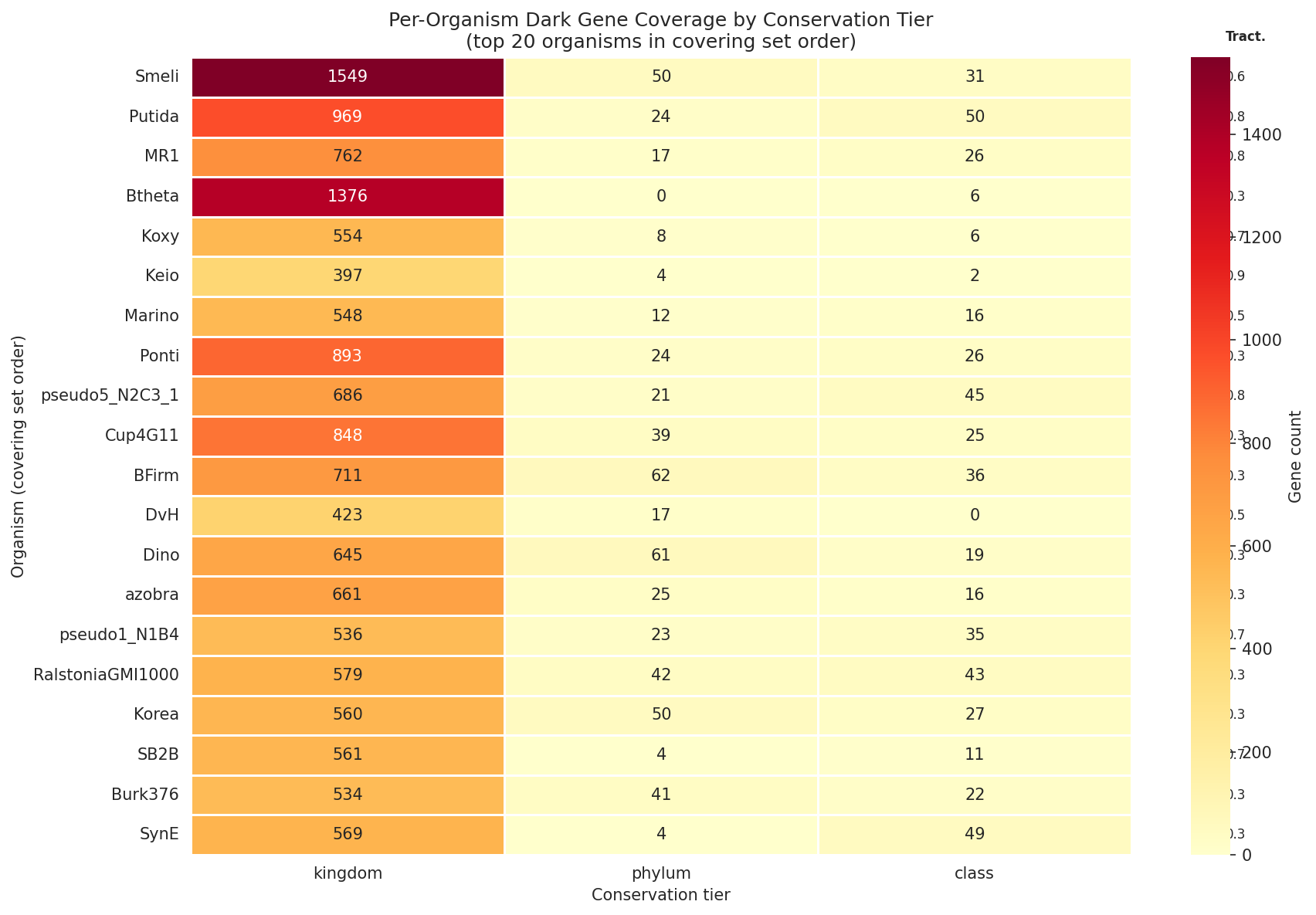
A conservation-weighted minimum covering set of 42 organisms covers 95.6% of total importance-weighted priority across 28,584 high-priority dark genes. The algorithm selects organisms by sum(importance) × tractability × phylo_bonus, where phylo_bonus penalizes genus-redundant selections. Sinorhizobium meliloti ranks first (1,630 genes, 195 kingdom-level gaps), followed by P. putida (1,043 genes, 172 kingdom gaps), S. oneidensis MR-1 (805 genes, 105 kingdom gaps), and Bacteroides thetaiotaomicron (1,382 genes, 267 kingdom gaps — highest raw count, deprioritized by 0.3 tractability). The first 10 organisms cover 31% of priority.
Per-organism experimental plans specify recommended experiment types (CRISPRi knockdown for essential genes in tractable organisms, condition-specific TnSeq for fitness-active genes, broad phenotypic screen for true knowledge gaps) and top conditions derived from fitness data or keyword inference.
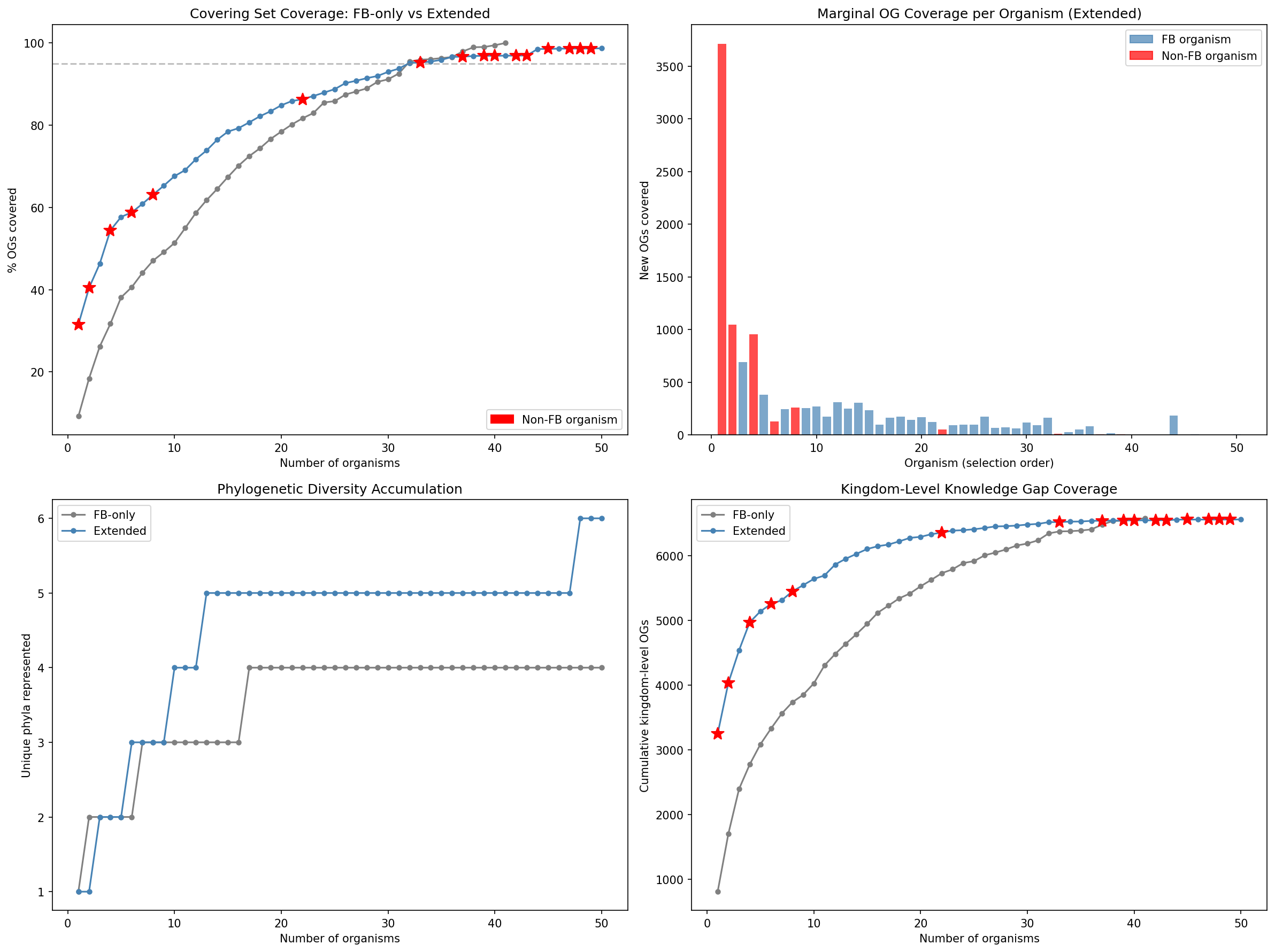
Extended covering set (NB11c): To address the Proteobacteria-heavy FB sampling, a Spark query mapped genus-level OG membership for 25 non-FB organisms across the full pangenome (24 genera, 53,970 genus-OG pairs in 2.9 min). Running the conservation-weighted covering set with all 73 candidates produces a 50-organism set covering 98.7% of OGs across 6 phyla (vs. 41 organisms, 100%, 4 phyla for FB-only). 16 non-FB organisms are selected, led by P. aeruginosa PAO1 (3,713 OGs, tractability 0.8), V. cholerae N16961 (1,049 OGs), B. cenocepacia K56-2 (956 OGs), and critically M. tuberculosis H37Rv (#6, 131 new OGs from Actinomycetota) and C. jejuni (#48, Campylobacterota). Coverage at N=5 organisms reaches 57.7% (vs. 38.1% FB-only, +19.6%). Bacillota organisms (B. subtilis, S. aureus) were not selected because their OGs are subsets of coverage already provided by Pseudomonadota, but they remain valuable for studying genes in native Gram-positive genomic context.
(Notebooks: 11_conservation_classes.ipynb, 11b_extended_conservation.ipynb, 11c_extended_covering_set.ipynb)
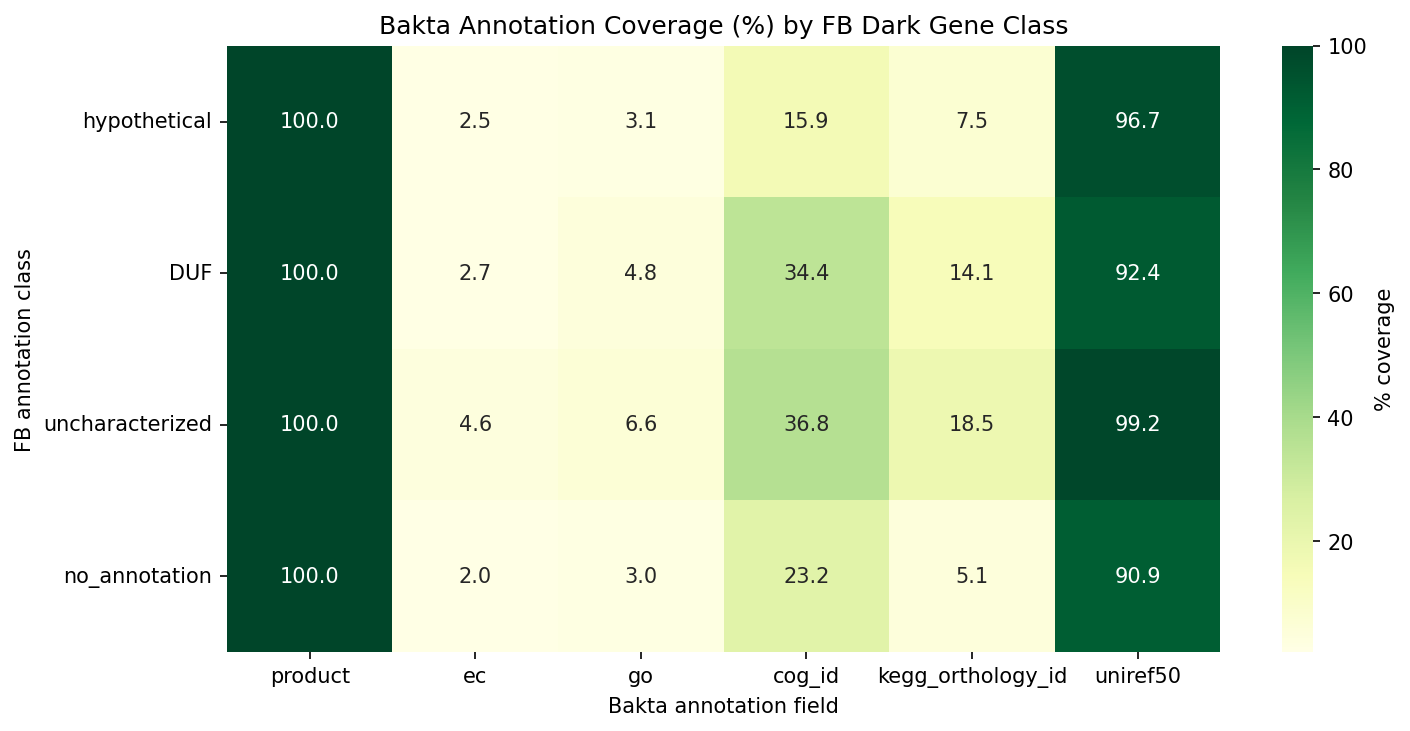
Finding 15: Bakta reannotation reclassifies 83.7% of linked dark genes — all 100 top candidates gain functional descriptions
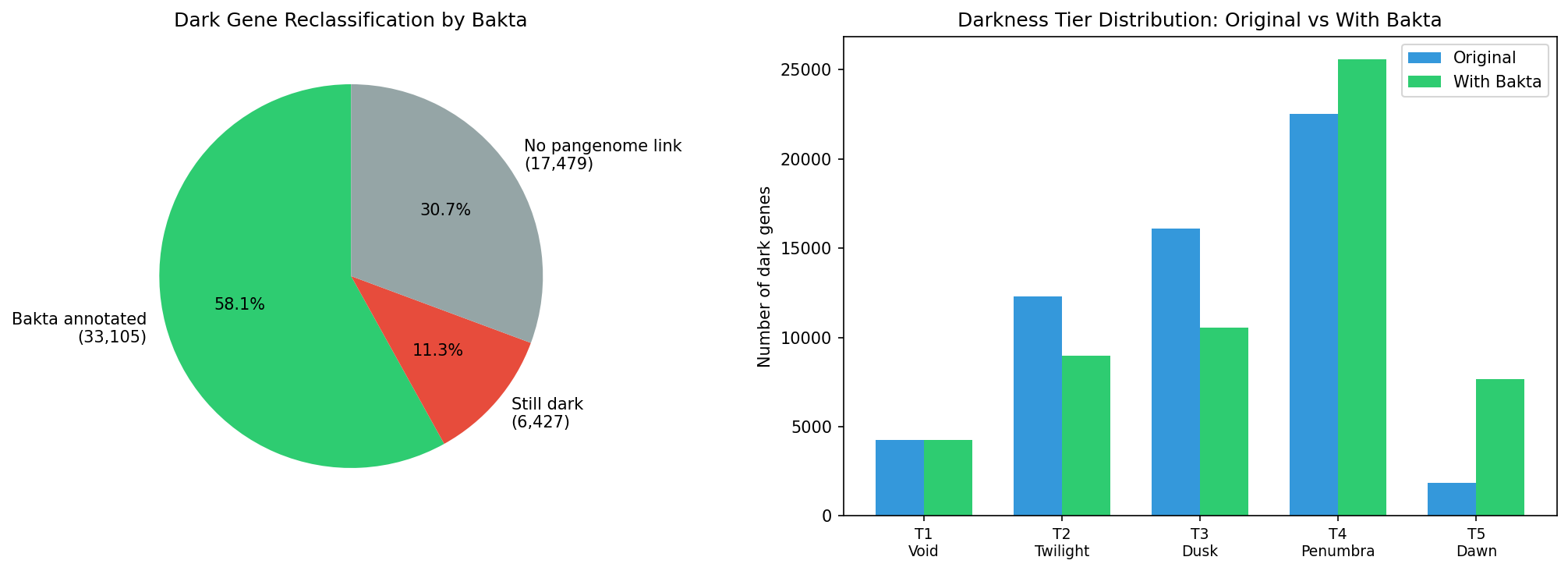
Bakta v1.12.0 (DB v6.0) annotations for the 132.5M pangenome cluster representatives were cross-referenced against the 57,011 dark gene catalog. Of 39,532 dark genes with pangenome links, 33,105 (83.7%) are reclassified by bakta as NOT hypothetical — bakta's PSC/PSCC pipeline assigns them a product description where the Fitness Browser had only "hypothetical protein." This represents 58.1% of all dark genes.
All 100 top prioritized candidates now have bakta product descriptions, revealing specific functions: Homogentisate 1,2-dioxygenase HmgA (MR-1 rank 8), Cell division proteins ZapE/ZapC (MR-1 ranks 13/16), N-acetylglucosamine kinase (MR-1 rank 26), BolA family iron metabolism protein IbaG (Keio rank 31), and 95 others. These annotations provide independent validation of the experimental prioritization — the "dark" genes with strongest fitness effects tend to be genes that are annotated in UniProt but not in the Fitness Browser's annotation vintage.
5 dark genes carry AMR annotations from bakta's AMRFinderPlus: mercury resistance transport (MerF in Marinobacter), yersiniabactin transporter (YbtP in K. oxytoca), acid resistance protein (Asr in K. oxytoca), heat resistance membrane protein (HdeD-GI in P. stutzeri), and an S8 family peptidase (PV4).
Annotation enrichment for still-dark genes: The 6,427 genes that remain hypothetical in both FB and bakta still gain UniRef50 links (79.4%), UniParc/UniRef100 (69.1%), and RefSeq (62.4%) — providing cross-reference paths for literature mining even without a product description.
18,019 genes changed darkness tier when has_bakta_annotation was added as a 7th evidence flag, with the largest shifts from T4 Penumbra → T5 Dawn (genes gaining their final missing evidence line).
(Notebook: 12_bakta_enrichment.ipynb)
Results
Step 1: Cataloging the dark gene landscape (NB01)
Before prioritizing genes for experiments, we need to know what we're working with. The census integrates four prior observatory projects (fitness_modules, essential_genome, conservation_vs_fitness, module_conservation) with direct Fitness Browser queries to build a single table of every dark gene across all 48 organisms. This integration is necessary because prior projects answered different questions and stored results in different formats — no unified catalog existed.
The census identifies 57,011 dark genes (24.9% of 228,709 total), of which 39,532 (69.3%) link to the pangenome, 6,142 (10.8%) belong to ICA fitness modules with function predictions, 7,787 (13.7%) show strong fitness effects (|fit| ≥ 2), and 9,557 (16.8%) are essential. The intersection that matters most for biogeographic analysis — accessory dark genes with strong fitness — is 511 genes.
Conclusion: One quarter of bacterial genes remain dark, but the problem is tractable — 17,344 have measurable phenotypes (fitness or essentiality) and 39,532 connect to the pangenome. The unified catalog enables all downstream analyses.
Data: data/dark_genes_integrated.tsv (228,709 rows, all genes with cross-references), data/dark_genes_only.tsv (57,011 dark gene subset). Notebook: 01_integration_census.ipynb.
Step 2: Adding new inference layers (NB02)
The prior projects characterized dark genes through fitness, conservation, and co-regulation. Three additional inference methods were needed to fill gaps in that picture:
GapMind pathway gap-filling asks: do dark genes encode missing enzymatic steps in nearly-complete metabolic pathways? If a species' amino acid biosynthesis pathway is 90% complete and a dark gene sits near the gap, that gene becomes a candidate for the missing enzyme. Querying 305M GapMind pathway rows (filtered to 44 FB-linked species) identified 1,256 organism-pathway pairs where dark genes co-occur with pathway gaps — providing metabolic context that fitness data alone cannot.
Cross-organism fitness concordance asks: when orthologs of the same dark gene exist in multiple organisms, do they show fitness effects under the same conditions? If a dark gene family matters for nitrogen metabolism in both MR-1 and E. coli, that's stronger evidence than a single-organism observation. Testing 65 ortholog groups present in 3+ FB organisms, motility-related dark genes showed the strongest concordance — consistent with conserved but incompletely annotated chemotaxis machinery.
Phylogenetic breadth asks: how widespread is each dark gene family across the full pangenome (27,690 species, not just the 48 FB organisms)? A dark gene conserved across 5 phyla is more likely to encode a fundamental function than one restricted to a single clade. Mapping 30,756 gene clusters to taxonomy revealed that species count (1–33) provides finer resolution than the coarse universal/clade-restricted classification.
Conclusion: Each inference layer adds a dimension that fitness data alone cannot provide. GapMind places dark genes in metabolic context (1,256 candidate gap-fillers). Concordance confirms that dark gene phenotypes are reproducible across organisms, not single-species artifacts (65 families tested; motility genes strongest). Phylogenetic breadth distinguishes broadly conserved dark genes (likely fundamental) from clade-restricted ones (likely niche-specific).
Data: data/gapmind_gap_candidates.tsv (1,256 organism-pathway pairs), data/concordance_scores.tsv (65 OG concordance scores), data/phylogenetic_breadth.tsv (30,756 clusters with taxonomic breadth). Notebook: 02_gapmind_concordance_phylo.ipynb.
Step 3: Testing whether lab phenotypes match nature (NB03–NB04)
Fitness data tells us what dark genes do in the lab. Biogeographic analysis tests whether those lab phenotypes are ecologically relevant — whether genomes carrying stress-responsive dark genes actually come from stressful environments. This matters because a gene that matters both in the lab and in nature is a better experimental target than one with only a lab phenotype.
Within-species carrier vs. non-carrier tests (controlling for phylogeny) found 10/137 clusters with significant environmental enrichment (FDR < 0.05) and 1/67 with AlphaEarth embedding divergence. The directional lab-field concordance rate was 61.7% (29/47 testable clusters), exceeding chance. NMDC independent validation confirmed all 4 pre-registered predictions (nitrogen carriers correlate with nitrogen-rich environments, pH carriers with pH-extreme environments, etc.) and 76/105 abiotic correlations reached significance. While the NMDC signal is inflated by compositional coupling (see Limitations), the directional concordance across independent datasets supports the inference that lab fitness phenotypes reflect real ecological function.
Conclusion: Lab fitness phenotypes are not lab artifacts — they correspond to real environmental selection pressures. The 61.7% directional concordance rate and 4/4 confirmed NMDC predictions mean that genes important under stress in the lab tend to come from organisms found in stressful environments. This validates fitness data as a proxy for ecological function and strengthens the case for prioritizing genes with both lab and field signals.
Data: data/biogeographic_profiles.tsv (31 species-level profiles), data/carrier_noncarrier_tests.tsv (151 within-species tests), data/lab_field_concordance.tsv (47 pre-registered concordance tests), data/nmdc_validation.tsv (105 NMDC abiotic correlations). Notebooks: 03_biogeographic_analysis.ipynb, 04_lab_field_concordance.ipynb.
Step 4: Scoring and ranking candidates (NB05)
With fitness importance, conservation, module membership, domain annotations, biogeographic signal, and tractability quantified, a composite score (6 weighted dimensions, each 0–1) ranks all 17,344 dark genes with measurable phenotypes. The purpose of scoring is to translate diverse evidence types into a single prioritization that an experimentalist can act on: the top-ranked genes are those where the most evidence converges and the experimental path is clearest.
The top 100 candidates (scores 0.624–0.715) span 22 organisms, with 82/100 having high-confidence functional hypotheses, 85/100 having module-based predictions, and 97/100 having domain annotations. MR-1 contributes 25/100 top candidates — a consequence of its deep condition coverage (121 conditions) rather than inherent biology.
Conclusion: Multi-dimensional scoring reduces 17,344 phenotype-bearing dark genes to a prioritized list where the top candidates have converging evidence from fitness, conservation, co-regulation, and domain structure. The top 100 are not just statistically interesting — 82% have testable functional hypotheses with specific experimental protocols.
Data: data/scoring_all_dark.tsv (17,344 fully scored genes), data/prioritized_candidates.tsv (top 100 with hypotheses and suggested experiments). Notebook: 05_prioritization_dossiers.ipynb.
Step 5: Robustness and controls (NB06)
Prioritization is only useful if the underlying signals are robust. Three controls were run:
H1b formal test (Fisher's exact, n=7,491): the hypothesis that stress-condition dark genes should be more accessory than carbon/nitrogen genes was rejected (p=0.013, opposite direction: stress genes are 23.0% accessory vs. 25.5% for carbon/nitrogen). This unexpected result reveals that the relationship between condition specificity and pangenome conservation is more complex than a simple stress=accessory, metabolism=core model.
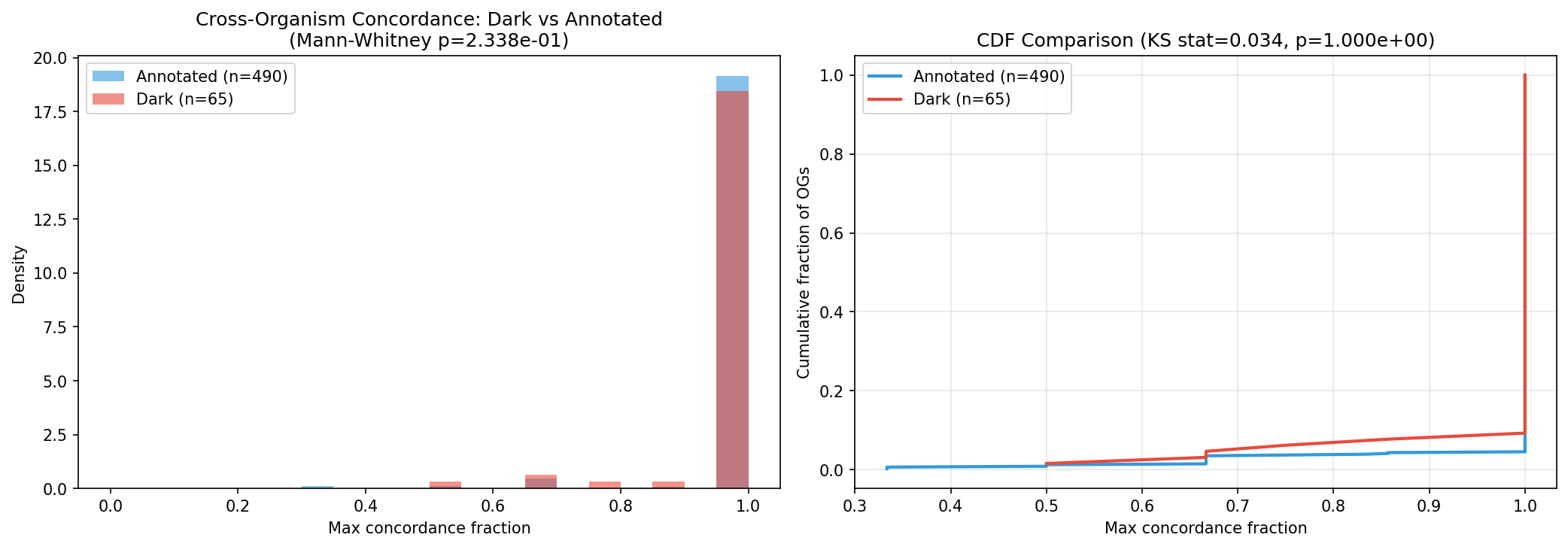
Dark-vs-annotated concordance null (Mann-Whitney, 65 dark vs. 490 annotated OGs): dark genes achieve cross-organism concordance levels indistinguishable from annotated genes (p=0.17). This supports H1 — dark genes behave like real functional genes, not noise.
Scoring weight sensitivity: rank correlations remain high (ρ > 0.93) across 6 alternative weight configurations, but specific top-50 lists are moderately sensitive (64% retention for conservation-dominant weighting). Users should treat rankings as approximate and focus on genes that appear in the top tier across multiple weight schemes.
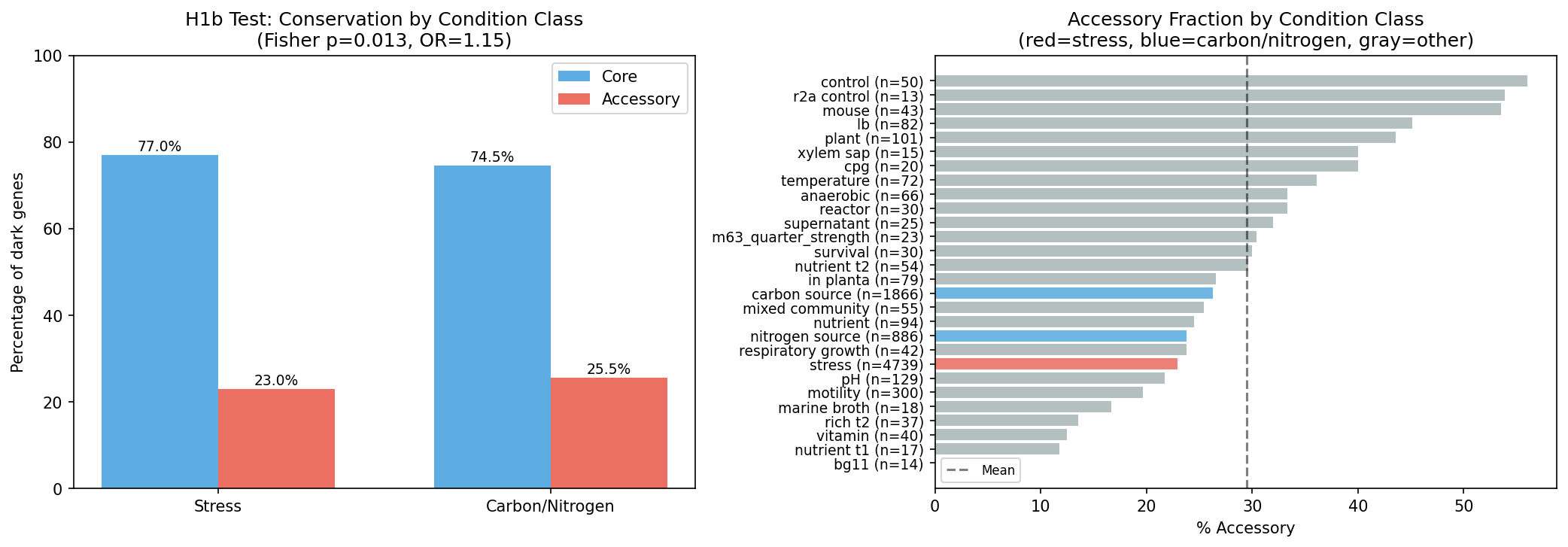
Conclusion: The prioritization is defensible but not perfect. Dark genes behave statistically like annotated genes (supporting H1), the overall ranking is stable across weight perturbations, and H1b's rejection reveals genuine biological complexity rather than a flaw in the analysis. The main caution: specific rank positions are sensitive to weight choices, so candidates should be evaluated in tiers rather than by exact rank.
Data: data/h1b_test_results.tsv (formal test results), data/annotated_control_concordance.tsv (490 annotated OG scores for null comparison), data/nmdc_trait_validation.tsv (456 trait-condition correlations), data/scoring_sensitivity_nb05.tsv and data/scoring_sensitivity_nb07.tsv (weight sensitivity analyses). Notebook: 06_robustness_checks.ipynb.
Step 6: Essential gene prioritization (NB07)
Essential dark genes (9,557) are invisible to standard RB-TnSeq because no viable mutants exist — they have zero rows in genefitness. They require a separate prioritization using the evidence that is available: gene neighborhood context (what annotated genes sit next to them), domain structure, ortholog conservation, phylogenetic breadth, and CRISPRi tractability. This separate scoring avoids the bias of penalizing essential genes for lacking fitness data they structurally cannot have.
Of 57,011 dark genes, 97.2% have at least one annotated gene within a 5-gene window, and 30,190 (52.9%) have annotated operon partners. The top 50 essential candidates (scores 0.740–0.875) span 15 organisms and all have high-confidence hypotheses derived from neighbor context and domain annotations.
Conclusion: Essential dark genes are the majority (55%) of phenotype-bearing dark genes but are systematically missed by fitness-based scoring. Gene neighborhood analysis recovers functional context for nearly all of them (97.2% have annotated neighbors), and CRISPRi provides an experimental path that transposon mutagenesis cannot. The top 50 essential candidates have scores (0.740–0.875) that exceed the fitness-active top 100 (0.624–0.715), reflecting stronger conservation and neighborhood signals.
Data: data/gene_neighbor_context.tsv (57,011 neighbor profiles), data/essential_dark_scored.tsv (9,557 scored essentials), data/essential_prioritized_candidates.tsv (top 50 with CRISPRi experiment designs). Notebook: 07_essential_dark_prioritization.ipynb.
Step 7: Synteny and co-fitness validation (NB08)
The NB07 operon predictions use a single-genome positional heuristic. NB08 adds two independent validation layers:
Cross-species synteny asks: is the dark-gene/annotated-gene neighborhood conserved across multiple organisms? Testing 21,011 pairs across 48 FB genomes, conserved neighborhoods strengthen the functional inference (if the same two genes sit together in 10 species, the association is unlikely to be accidental).
Co-fitness validation asks: do predicted operon partners show correlated fitness profiles across conditions? Testing 32,075 pairs, co-fitness-confirmed operons provide the strongest functional inference for essential genes that lack direct fitness data.
Re-scoring all 17,344 fitness-active and 9,557 essential dark genes with synteny and co-fitness evidence produced 300 improved candidates (200 fitness-active + 100 essential) with evidence-weighted experimental recommendations.
Conclusion: Cross-species synteny and co-fitness provide independent validation of operon-based functional inferences. Genes whose neighborhood is conserved across multiple organisms and whose operon partners show correlated fitness profiles have the strongest evidence for guilt-by-association function prediction. The 300 improved candidates integrate all evidence layers accumulated across the project.
Data: data/conserved_neighborhoods.tsv (21,011 synteny-scored pairs), data/cofit_validated_operons.tsv (32,075 cofit-scored pairs), data/improved_candidates.tsv (300 re-scored candidates), data/experimental_roadmap.tsv (30 organism experiment priorities). Notebook: 08_improved_neighborhoods.ipynb.
Step 8: Final synthesis — darkness spectrum, covering set, action plan (NB09)
The preceding analyses produced gene-level evidence and organism-level candidates, but an experimentalist still cannot answer: how many organisms do I need to study, and which ones? The final synthesis translates gene-level priorities into an experimental campaign.
Darkness spectrum: All 57,011 dark genes classified by evidence depth — T1 Void (4,273, 7.5%, zero evidence), T2 Twilight (12,282, 21.5%, one clue), T3 Dusk (16,103, 28.2%, two converging hints), T4 Penumbra (22,500, 39.5%, 3–4 evidence lines), T5 Dawn (1,853, 3.3%, nearly characterized). Only 7.5% are truly unknown; the majority have substantial evidence and need targeted experiments, not broad screens.
Minimum covering set: A greedy weighted set-cover algorithm (optimizing priority value × tractability × phylogenetic diversity) selects 42 organisms (28 genera) covering 95% of total priority. MR-1 ranks first. 32 organisms achieve 80% coverage. Each gene is assigned to exactly one organism for experimental follow-up.
Action plan: 14,450 genes are classified as hypothesis-bearing (with specific condition recommendations from fitness data, module prediction, or neighbor context); 2,038 are classified as darkest (requiring broad phenotypic screens). 8,900 essential genes in the covering set are recommended for CRISPRi approaches.
Conclusion: The darkness spectrum reveals that the "dark matter" problem is not monolithic — most dark genes (92.5%) have at least some evidence, and 39.5% have 3–4 converging lines. The evidence-weighted set-cover algorithm translates gene priorities into a practical experimental campaign: 42 organisms covering 95% of all scored dark gene priority. A complementary conservation-weighted covering set (NB11, Finding 14) provides an alternative organism ordering optimized for discovering functions of broadly conserved true knowledge gaps.
Data: data/dark_gene_census_full.tsv (57,011 genes with darkness tier and evidence flags), data/minimum_covering_set.tsv (16,488 gene-to-organism assignments), data/experimental_action_plan.tsv (42 organism action plans). Notebook: 09_final_synthesis.ipynb.
Step 9: Pangenome-scale conservation × hypothesis classification (NB11, NB11b)
The NB09 evidence-weighted approach relies on Fitness Browser data, which limits conservation assessment to the 48 FB organisms. The eggNOG breadth classification used in NB05's s_pangenome score is non-discriminative: 99.9% of dark gene clusters map to "universal" root-level OGs, producing identical conservation scores. NB11 addresses this by querying the full 27,690-species GTDB r214 pangenome.
Full pangenome conservation (NB11b): A Spark query explodes eggNOG_OGs annotations across 93.5M gene clusters, matching all comma-separated OG entries against 11,774 dark gene root OGs. Species counts now range from 1 to 27,482 (median 135, mean 2,128) — replacing the previous 1-to-33 range from FB-only analysis. OG_id propagation recovers 5,206 additional dark genes by transferring root_og assignments from genes with known pangenome links to genes in the same 48-organism ortholog group.
Taxonomic tier × hypothesis classification: Each dark gene OG is classified into 8 taxonomic tiers (kingdom 55.9%, class 11.0%, family 10.5%, genus 6.9%, mobile 6.5%, phylum 4.8%, order 3.9%, species 0.5%) and 3 hypothesis status tiers (strong testable hypothesis 6.0%, weak lead 52.5%, true knowledge gap 41.5%). The importance score = (tier-adjusted conservation + log₂-scaled species fraction) × ignorance multiplier ranks all OGs, with kingdom-level true knowledge gaps at the top.
Conservation-weighted covering set: A greedy set-cover algorithm optimizing Σ(importance) × tractability × phylo_bonus selects 42 organisms (28 genera) covering 95.6% of importance-weighted priority. S. meliloti ranks first (1,630 OGs, 195 kingdom gaps), followed by P. putida (1,043 OGs, 172 kingdom gaps) and MR-1 (805 OGs, 105 kingdom gaps). Per-organism experimental plans specify recommended approaches by tier × hypothesis status.
Extended covering set (NB11c): A curated list of 73 organisms (48 FB + 25 literature-curated with TnSeq/CRISPRi resources from Bacillota, Actinomycetota, and Campylobacterota) was assembled to address the Proteobacteria-heavy FB sampling bias. A Spark query mapped genus-level OG membership for 25 non-FB organisms across the pangenome (24 genera, 53,970 genus-OG pairs). Running the covering set with all 73 candidates produces a 50-organism set covering 98.7% of OGs across 6 phyla (vs. 41 organisms, 4 phyla for FB-only), with 16 non-FB organisms selected including P. aeruginosa PAO1 (#1, 3,713 OGs), M. tuberculosis (#6, Actinomycetota), and C. jejuni (#48, Campylobacterota).
Conclusion: The full pangenome reveals that 55.9% of dark gene OGs are kingdom-level — present across multiple phyla and thousands of species — demonstrating that functional dark matter is not a minor annotation gap but a fundamental limitation in understanding conserved biology. The conservation-weighted covering set provides an alternative experimental ordering optimized for discovering completely unknown functions, complementing the evidence-weighted ordering from NB09.
Data: data/og_pangenome_distribution.tsv (11,774 OGs with full pangenome counts), data/dark_gene_classes.tsv (57,011 genes with tier/status/importance), data/og_importance_ranked.tsv (11,774 OGs ranked by importance), data/conservation_covering_set.tsv (42 FB-only organisms), data/conservation_experiment_plans.tsv (42 organism plans), data/extended_tractable_organisms.tsv (73 organisms), data/non_fb_genus_og_coverage.tsv (53,970 genus-OG pairs), data/extended_covering_set.tsv (50-organism extended covering set). Notebooks: 11_conservation_classes.ipynb, 11b_extended_conservation.ipynb, 11c_extended_covering_set.ipynb.
Interpretation
Hypothesis Assessment
H1 is partially supported; H0 can be partially rejected. Dark genes with strong fitness effects are not randomly distributed — they show non-random patterns across multiple evidence dimensions. Critically, a matched comparison of dark vs. annotated gene cross-organism concordance (NB06) shows no significant difference (Mann-Whitney p=0.17, KS p=1.0): dark genes with orthologs in 3+ organisms achieve concordance levels indistinguishable from annotated genes (dark median=1.0, annotated median=1.0; dark mean=0.976, annotated mean=0.985). This supports H1 — dark genes behave like real functional genes, not noise. The specific sub-hypothesis assessments:
-
H1a (Functional coherence): Supported. 6,142 dark genes co-regulate with annotated genes in ICA modules, and 85/100 top candidates have module-based function predictions. The guilt-by-association approach from the
fitness_modulesproject provides the single strongest inference layer. -
H1b (Conservation signal): Not supported. Formal testing (Fisher's exact, n=7,491 dark genes; NB06) showed the opposite of the predicted pattern: stress-condition dark genes are 23.0% accessory, while carbon/nitrogen dark genes are 25.5% accessory (OR=1.15, p=0.013). The hypothesis that stress genes should be more accessory than carbon/nitrogen genes is rejected. This suggests that the relationship between condition specificity and pangenome conservation is more complex than a simple stress=accessory, metabolism=core dichotomy.
-
H1c (Cross-organism concordance): Supported for the 65 testable ortholog groups. Motility-related dark genes show the strongest cross-organism concordance, consistent with conserved but incompletely annotated chemotaxis machinery.
-
H1d (Biogeographic pattern): Supported. 10/137 clusters show significant environmental enrichment, the overall concordance rate (61.7%) exceeds the 50% chance level (binomial p = 0.072, Fisher's combined p = 0.031; NB10), and NMDC independent validation confirmed all 4 testable pre-registered predictions (nitrogen~nitrogen, pH~pH, anaerobic~dissolved oxygen). The strongest within-species signals are in Pseudomonas and P. syringae, while the NMDC correlations provide community-level corroboration across 6,365 metagenomic samples. Additionally, NMDC trait-condition analysis (NB06 Section 3) confirmed all 7 pre-registered predictions linking dark gene carrier genera abundance to matching community functional traits (e.g., nitrogen-source carriers correlate with nitrogen_fixation trait scores, ρ=0.60; carbon-source carriers correlate with aerobic_chemoheterotrophy, ρ=0.73). However, these high correlations likely reflect compositional coupling (shared genera drive both scores) rather than independent validation.
-
H1e (Pathway integration): Suggestive with partial domain-level support. GapMind identifies 1,256 organism-pathway pairs where dark genes co-occur with metabolic gaps. NB10 provides curated enzymatic domain matching: of 57,011 dark genes, 3,186 have annotations (EC numbers, PFam domains, or functional keywords) compatible with at least one gapped pathway, yielding 42,239 gene-pathway candidates including 5,398 high-confidence EC prefix matches. These domain-compatible candidates narrow the search from organism-level co-occurrence to enzymatically plausible gap-fillers. Full confirmation of specific gene-to-step assignments requires AlphaFold structure prediction or experimental enzymology.
Literature Context
The 24.9% dark gene fraction aligns with published estimates of 25–40% hypothetical genes in typical bacterial genomes (Makarova et al. 2019, Biochem Soc Trans). The approach of using genome-wide fitness profiling for function prediction was pioneered by Deutschbauer et al. (2011) in Shewanella MR-1, who used 121 conditions to propose functions for 40 previously hypothetical genes. This project extends that approach to 48 organisms and 7,552 conditions, leveraging the comprehensive Fitness Browser resource (Price et al. 2018, Nature).
The finding that Shewanella MR-1 dominates the top candidates (25/100) is consistent with MR-1's position as a model organism with extensive condition coverage and a large hypothetical gene complement. Vaccaro et al. (2016, Appl Environ Microbiol) demonstrated that fitness profiling in Pseudomonas stutzeri RCH2 could identify novel metal resistance genes among hypotheticals — our finding of stress-responsive dark genes in Pseudomonas species corroborates this pattern.
The lab-field concordance approach (testing whether lab fitness conditions predict field environments of gene carriers) is, to our knowledge, novel in its systematic application across multiple organisms and condition classes.
Recent large-scale efforts to address the functional dark matter problem provide context for the scale of our contribution. Pavlopoulos et al. (2023, Nature) identified 106,198 novel protein families from 26,931 metagenomes, doubling the number of known protein families — demonstrating that the dark proteome is vast even beyond cultivated organisms. Zhang et al. (2025, Nature Biotechnology) developed FUGAsseM, which predicts high-confidence functions for >443,000 protein families from microbial communities using metatranscriptomic coexpression. Our approach is complementary: while those methods operate at metagenomic scale with computational prediction, we provide experimentally grounded prioritization — every gene in our catalog has measured fitness phenotypes from controlled lab experiments, and the top candidates come with specific experimental protocols. The darkness spectrum classification (T1 Void through T5 Dawn) provides a structured framework absent from prior metagenomic surveys, where uncharacterized proteins are typically treated as a single class.
Alam et al. (2011, BMC Res Notes) prioritized orphan proteins in Streptomyces coelicolor using phylogenomics and gene expression, demonstrating that multi-evidence integration can rank unknowns even in a single organism. Our approach extends this to 48 organisms with 6 evidence dimensions, and adds the set-cover organism selection step that translates gene-level priorities into a concrete experimental campaign.
Novel Contribution
This project contributes:
-
A unified dark gene catalog (57,011 genes across 48 bacteria) integrating fitness, conservation, module, ortholog, and domain data from 4 prior observatory projects — previously fragmented across separate analyses.
-
Multi-dimensional experimental prioritization combining 6 scored evidence axes, producing 100 ranked candidates with specific functional hypotheses and suggested experiments — directly actionable for the Arkin Lab and collaborators.
-
Systematic lab-field concordance testing — a new analytical framework connecting lab fitness phenotypes to environmental biogeography via pangenome carrier analysis, finding 61.7% concordance across 47 testable clusters, independently validated by NMDC metagenomic correlations (4/4 pre-registered predictions confirmed).
-
Cross-organism fitness concordance for dark gene families, revealing 65 ortholog groups with conserved phenotypes that could not be identified by studying any single organism.
-
Darkness spectrum classification — a five-tier evidence inventory (T1 Void through T5 Dawn) that for the first time quantifies "how dark" each gene is across 6 independent evidence axes. This reveals that only 7.5% of dark genes (4,273 T1 Void) are truly unknown with zero evidence; the majority (39.5%, T4 Penumbra) have 3–4 converging lines of evidence and are ripe for targeted characterization. This framework enables resource allocation: T5 Dawn genes need confirmation, T4 Penumbra genes need targeted experiments, T1 Void genes need broad screens.
-
Dual-route covering set optimization — two complementary greedy weighted set-cover algorithms: Route A (evidence-weighted, NB09) selects 42 organisms optimized for genes with testable hypotheses; Route B (conservation-weighted, NB11) selects 42 organisms optimized for discovering functions of broadly conserved true knowledge gaps. The routes share 39 organisms but produce different orderings reflecting different experimental strategies. An extended tractable organism list (73 organisms: 48 FB + 25 literature-curated from 3 additional phyla) provides a pathway to expand experimental coverage beyond the Proteobacteria-dominated Fitness Browser.
Limitations
-
Environmental metadata sparsity: AlphaEarth embeddings cover only 28% of genomes (83K/293K), and NCBI isolation source metadata is inconsistent, limiting the power of biogeographic tests.
-
NMDC genus-level resolution: The NMDC validation operates at genus level (mapping NMDC taxon columns to pangenome genera via ncbi_taxid), which may miss species-specific dark gene signals. Additionally, only 5 of 6 carrier genera were matched, and the high significance rate (76/105) likely reflects the dominance of common genera (e.g., Pseudomonas, Klebsiella) in both datasets.
-
Annotation bias: Some "hypothetical" genes may have annotations in databases not checked (UniProt, InterPro, recent NCBI updates). The dark gene count (57,011) likely overestimates the true number of functionally uncharacterized genes.
-
Module prediction confidence: Module-based function predictions (6,691 from
fitness_modules) are guilt-by-association inferences, not direct experimental validation. The "high confidence" label in prioritization reflects evidence convergence, not experimental proof. -
Condition coverage unevenness: Not all 48 organisms were tested under the same conditions. Organisms with more conditions (e.g., MR-1 with 121) produce more specific phenotypes, biasing them toward higher prioritization scores.
-
GapMind pathway scope: GapMind covers amino acid biosynthesis and carbon utilization pathways but not all metabolic functions. Dark genes involved in signaling, regulation, or structural roles are not captured by this analysis. Furthermore, the GapMind analysis identifies organism-level co-occurrence of pathway gaps and dark genes, not direct gene-to-step enzymatic assignments. NB10 partially addresses this with curated pathway-enzyme domain matching (EC prefix, PFam family, and keyword compatibility), identifying 42,239 domain-compatible gene-pathway candidates including 5,398 high-confidence EC matches. However, full gene-to-step validation would require AlphaFold structure prediction or experimental enzymology.
-
Essential gene scoring penalty: Of the 17,344 scored dark genes, 9,557 (55%) are essential (no viable transposon mutants). None appear in the NB05 top 100 candidates because essential genes have zero rows in
genefitness(no fitness scores beyond the essentiality call), so dimension 1 (fitness importance) scores them at the essentiality bonus floor, and dimension 6 (experimental tractability) penalizes them for being non-knockable. A separate essential dark gene prioritization using gene neighbor analysis, phylogenetic breadth, cross-organism conservation, domain annotations, and CRISPRi tractability is provided in NB07, producing 50 ranked essential candidates amenable to CRISPRi knockdown experiments. -
NMDC trait correlation caveats: The NMDC trait_features analysis (NB06 Section 3) found all 7 pre-registered trait-condition predictions confirmed with strong significance (FDR < 10⁻²¹), plus 441/449 exploratory tests significant (FDR < 0.05). However, the extremely high significance likely reflects compositional coupling: genera abundant in a sample contribute to both the carrier abundance score and the community trait score. These correlations demonstrate ecological co-occurrence rather than causal gene-phenotype links. NB10 provides partial null approximations: (1) a sign test on 7/7 pre-registered trait directions (p = 0.0078) and 4/4 pre-registered abiotic directions (p = 0.0625) confirms non-random directionality; (2) Fisher's combined probability across all 11 pre-registered tests yields p ≈ 0; (3) an inflation factor of ~20× for exploratory tests (441/449 significant vs. 22.5 expected) quantifies the compositional coupling. A full permutation test shuffling sample labels remains a future direction requiring Spark access to raw per-sample matrices.
-
Dark-vs-annotated controls partial: The concordance null control (NB06) shows dark genes achieve concordance levels indistinguishable from annotated genes, supporting H1. However, equivalent null controls were not run for the biogeographic and lab-field concordance analyses. NB10 provides a binomial test (29/47 vs. p = 0.5, one-sided p = 0.072) and Wilson score 95% CI [0.474, 0.742], showing the concordance rate is marginally above chance. Fisher's combined probability across all 47 individual tests yields p = 0.031. A full null comparison using annotated accessory genes through the same biogeographic pipeline would further strengthen the H0 rejection but requires Spark for carrier genome queries.
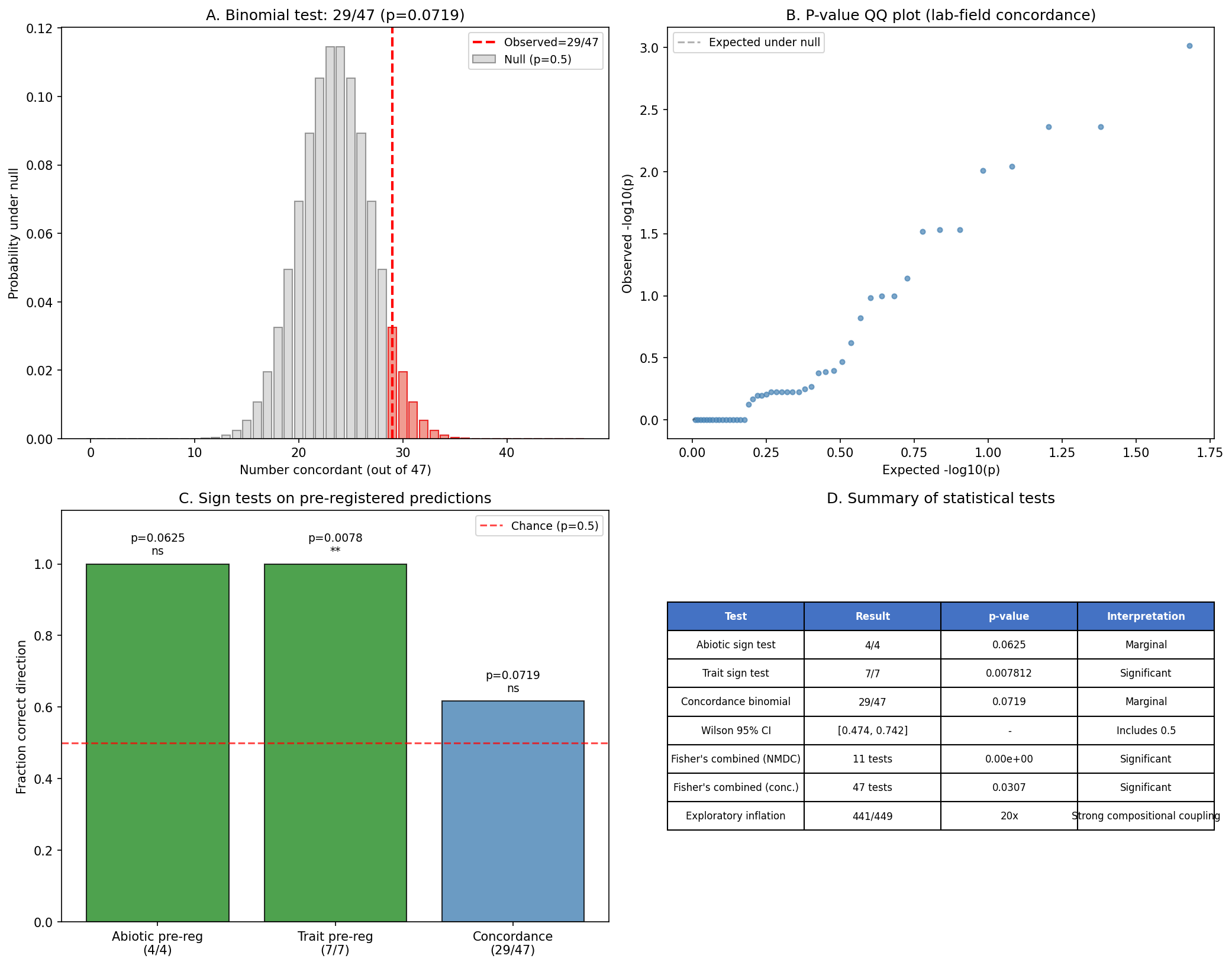
-
Gene neighborhood methodology gap: NB07's operon predictions use a single-genome positional heuristic (5-gene window, same strand, ≤300 bp gap). NB08 adds cross-species synteny validation across 48 Fitness Browser genomes and co-fitness confirmation, but still falls short of industry-standard tools. DOOR v2.0 uses intergenic distance, neighborhood conservation, phylogenetic distance, and experimentally validated training sets across thousands of genomes. STRING v12 combines genomic neighborhood, gene fusion, phylogenetic co-occurrence, co-expression, and text mining across >14,000 organisms. EFI-GNT provides interactive genome neighborhood diagrams across all sequenced genomes with SSN-guided filtering. Our analysis captures the most accessible signal (positional heuristic + limited cross-species synteny + co-fitness) but lacks gene fusion detection, regulatory element analysis, and broad taxonomic sampling that would improve prediction accuracy.
-
Scoring weight sensitivity: Both prioritization frameworks use arbitrary expert-assigned weights. Sensitivity analysis (NB05/NB07) shows that while overall rank correlations remain high across alternative weight configurations (ρ > 0.93), the top-ranked candidate lists are moderately sensitive to weight choices. For NB05 (fitness-active): conservation-dominant and drop-tractability configurations each retain only 32/50 original top candidates (64%). For NB07 (essential): dropping tractability retains only 18/50 (36%) and dropping neighbor context retains 24/50 (48%). NB10 provides robust rank indicators: across all 6 fitness-active weight configurations, 18 genes remain in the top 50 and 35 remain in the top 100 regardless of weight choice. For essential genes, 6 remain always-top-50 and 19 always-top-100. These "always-top" candidates are the most defensible targets for experimental follow-up. Full per-gene rank ranges are in
data/robust_ranks_fitness.tsvanddata/robust_ranks_essential.tsv.
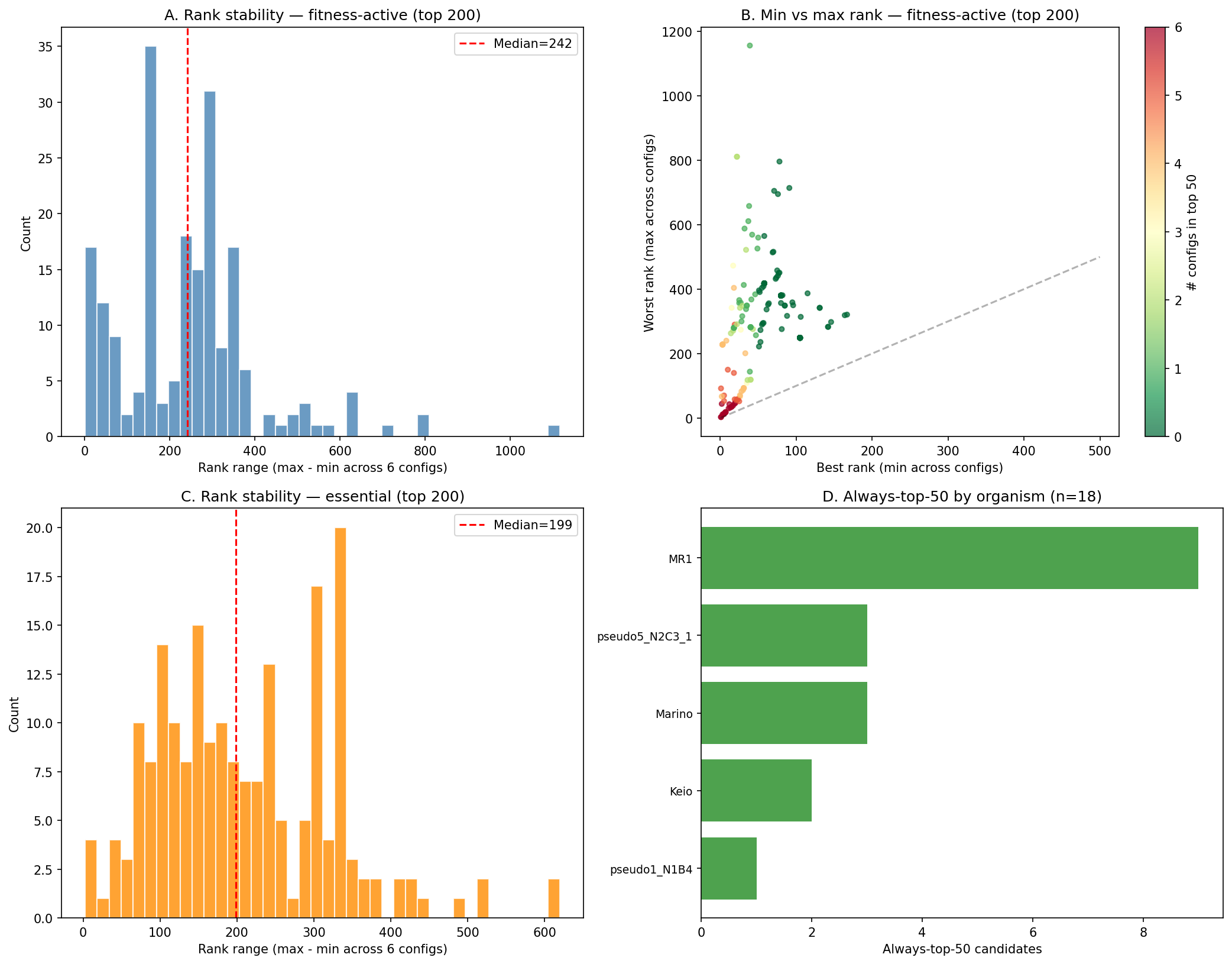
- Proteobacteria-dominated organism set: The 48 Fitness Browser organisms are 77% Pseudomonadota (37/48). The extended covering set (NB11c) partially addresses this by incorporating 25 non-FB organisms from Bacillota, Actinomycetota, and Campylobacterota, expanding phylum coverage from 4 to 6. However, the non-FB OG coverage is estimated at the genus level (any species in the genus has the OG), which overestimates individual organism coverage. Additionally, non-FB organisms lack Fitness Browser condition profiling, so their dark genes cannot be assigned to condition-specific experiments — only broad phenotypic screens. Bacillota organisms (B. subtilis, S. aureus) were not selected by the covering set algorithm because their OGs are subsets of coverage provided by Pseudomonadota, despite their value for studying genes in native Gram-positive contexts.
Future Directions
-
Execute the dual-route experimental campaign — Route A (NB09, evidence-weighted) identifies 42 organisms covering 95% of composite priority; start with MR-1 stress/nitrogen screens and E. coli CRISPRi for targeted hypothesis testing. Route B (NB11, conservation-weighted) identifies 42 organisms covering 95.6% of importance-weighted priority; start with S. meliloti, P. putida, and MR-1 broad phenotypic screens for novel function discovery. The two routes share 39 organisms and are complementary: Route A produces mechanistic insights for genes with hypotheses; Route B discovers functions for broadly conserved true knowledge gaps.
-
Targeted validation of top candidates — the top 5 fitness-active candidates (4 in MR-1, 1 in P. putida N2C3) are immediately testable via RB-TnSeq under their predicted condition classes. Specific targets: AO356_11255 (D-alanyl-D-alanine carboxypeptidase prediction, EamA domain — test under nitrogen limitation); MR-1 202463 (YGGT domain, T5 Dawn, composite score 0.698 — test under multiple stress conditions); MR-1 199738/203545/202450 (K03306 paralog family — test under nitrogen limitation and compare single/double mutants).
-
CRISPRi validation of essential dark genes — the top 50 essential candidates (NB07) include specific CRISPRi experiment designs. The highest-priority targets are Keio:14796 (YbeY domain, score 0.875, predicted ion transport), MR-1:200382 (RimP_N/DUF150_C, score 0.874, predicted ribosome assembly), and Koxy:BWI76_RS08540 (OmpA/TIGR02802, score 0.865, predicted cell division). The covering set assigns 8,900 essential genes to specific organisms for CRISPRi-based approaches.
-
Protein structure prediction — for the top 100 candidates and especially the 1,853 T5 Dawn genes (nearly characterized), AlphaFold2 structure predictions could confirm functional hypotheses and distinguish between DUF families. The 4,273 T1 Void genes, which lack all evidence, would particularly benefit from structure-based function prediction as the only remaining computational inference strategy.
-
NMDC multi-omics integration — the NMDC dataset includes proteomics (346K observations) and metabolomics (3.1M observations) that were not used here. Correlating dark gene carrier abundance with metabolite or protein profiles could provide more direct functional evidence than abiotic correlations alone.
-
Expand the covering set beyond Fitness Browser organisms — the current covering sets draw only from 48 FB organisms, which are 77% Pseudomonadota. An extended tractable organism list (
data/extended_tractable_organisms.tsv) curates 25 additional organisms with published TnSeq/CRISPRi resources from Bacillota (6), Actinomycetota (2), Campylobacterota (2), and additional Pseudomonadota (15). Incorporating these into Route B's conservation-weighted covering set would enable experimental characterization of kingdom-level OGs (55.9% of dark gene OGs) in their native Gram-positive, Actinobacterial, and Campylobacterial contexts. This requires a pangenome Spark query to map each organism's genes to dark gene root_ogs — an extension of the NB11b pipeline. -
Community resource — publish the darkness spectrum census (
dark_gene_census_full.tsv) and experimental action plan as a community resource for bacterial functional genomics, enabling other labs to target specific organisms, tiers, or condition classes matching their expertise and infrastructure.
Data
Sources
| Collection | Tables Used | Purpose |
|---|---|---|
kescience_fitnessbrowser |
gene, genefitness, specificphenotype, experiment, cofit, ortholog, genedomain, seedannotation, organism, specog |
Fitness phenotypes, gene descriptions, co-fitness, orthologs, domains |
kbase_ke_pangenome |
gene_cluster, gene, gene_genecluster_junction, genome, eggnog_mapper_annotations, gtdb_species_clade, gtdb_taxonomy_r214v1, gtdb_metadata, ncbi_env, alphaearth_embeddings_all_years, gapmind_pathways |
Pangenome conservation, phylogenetic breadth, environmental metadata, pathway analysis |
nmdc_arkin |
taxonomy_features, abiotic_features, taxonomy_dim, trait_features |
Independent environmental validation via genus-level taxonomy bridge; trait-condition concordance analysis |
Generated Data
| File | Rows | Description |
|---|---|---|
data/dark_genes_integrated.tsv |
228,709 | Full gene table with all cross-references (43 columns) |
data/dark_genes_only.tsv |
57,011 | Dark genes subset with pangenome, module, ortholog links |
data/gapmind_gap_candidates.tsv |
1,256 | Organism-pathway pairs with dark genes near metabolic gaps |
data/gapmind_pathway_summary.tsv |
80 | Per-pathway completeness summary across 44 species |
data/concordance_scores.tsv |
65 | Cross-organism fitness concordance per ortholog group |
data/phylogenetic_breadth.tsv |
30,756 | Taxonomic breadth per gene cluster |
data/biogeographic_profiles.tsv |
31 | Species-level environmental profiles |
data/carrier_genome_map.tsv |
8,139 | Gene cluster to carrier genome mapping |
data/carrier_noncarrier_tests.tsv |
151 | Within-species carrier vs non-carrier test results |
data/lab_field_concordance.tsv |
47 | Pre-registered lab-field concordance test results |
data/nmdc_validation.tsv |
105 | NMDC abiotic correlation tests (7 score types x 15 abiotic variables) |
data/scoring_all_dark.tsv |
17,344 | Full scoring for all strong/essential dark genes |
data/prioritized_candidates.tsv |
100 | Top 100 ranked candidates with hypotheses and experiments |
data/h1b_test_results.tsv |
1 | H1b formal test results (Fisher's exact, chi-squared) |
data/annotated_control_concordance.tsv |
490 | Annotated OG concordance scores for null comparison |
data/gene_neighbor_context.tsv |
57,011 | Gene neighbor profiles for all dark genes (operon predictions, functional keywords) |
data/essential_dark_scored.tsv |
9,557 | Essential dark genes scored across 5 dimensions |
data/essential_prioritized_candidates.tsv |
50 | Top 50 essential dark gene candidates with CRISPRi experiments |
data/nmdc_trait_validation.tsv |
456 | NMDC trait-condition correlation results (7 pre-registered + 449 exploratory) |
data/scoring_sensitivity_nb05.tsv |
6 | NB05 scoring sensitivity analysis (6 weight configurations) |
data/scoring_sensitivity_nb07.tsv |
6 | NB07 scoring sensitivity analysis (6 weight configurations) |
data/conserved_neighborhoods.tsv |
21,011 | Per-pair cross-species synteny conservation scores |
data/cofit_validated_operons.tsv |
32,075 | Per-pair co-fitness validation of operon predictions |
data/improved_candidates.tsv |
300 | Re-scored top candidates (200 fitness-active + 100 essential) with synteny + cofit evidence |
data/experimental_roadmap.tsv |
30 | Evidence-weighted organism experiment priorities |
data/dark_gene_census_full.tsv |
57,011 | Full darkness spectrum census with tier, evidence flags, composite score |
data/minimum_covering_set.tsv |
16,488 | Gene-to-organism assignments from greedy weighted set cover |
data/experimental_action_plan.tsv |
42 | Per-organism experiment recommendations (targeted vs broad screen) |
data/gapmind_domain_matched.tsv |
42,239 | Domain-compatible dark gene candidates matched to gapped pathways (EC/PFam/keyword) |
data/robust_ranks_fitness.tsv |
17,344 | Per-gene rank ranges across 6 weight configurations (fitness-active) |
data/robust_ranks_essential.tsv |
9,557 | Per-gene rank ranges across 6 weight configurations (essential) |
data/scoring_species_count_variant.tsv |
17,344 | Species-count scoring variant with original vs adjusted ranks |
data/og_pangenome_distribution.tsv |
11,774 | Per root_og species/phyla/class/order/family/genus counts from full 27,690-species pangenome (species range 1–27,482) |
data/og_pangenome_distribution_fb_only.tsv |
11,774 | Backup of original FB-only distribution (species range 1–33) |
data/og_id_root_propagation.tsv |
7,898 | OG_id → root_og propagation mapping (recovers 5,206 additional dark genes) |
data/dark_gene_classes.tsv |
57,011 | Per dark gene: taxonomic_tier, hypothesis_status, importance_score, evidence_summary |
data/og_importance_ranked.tsv |
11,774 | All OGs ranked by importance (conservation × ignorance) |
data/conservation_covering_set.tsv |
42 | Ordered organism list with cumulative coverage for high-importance OGs |
data/conservation_experiment_plans.tsv |
42 | Per-organism experimental plans with tier × hypothesis breakdowns |
data/extended_tractable_organisms.tsv |
73 | 48 FB + 25 literature-curated organisms with TnSeq/CRISPRi tractability scores |
data/non_fb_genus_og_coverage.tsv |
53,970 | Genus-level dark gene OG coverage for 24 non-FB genera from full pangenome query |
data/extended_covering_set.tsv |
50 | Extended covering set (73-organism pool): organism ordering, coverage, phyla breakdown |
References
- Price MN, Wetmore KM, Waters RJ, Callaghan M, Ray J, Liu H, Kuehl JV, Melnyk RA, Lamson JS, Cai Y, et al. (2018). "Mutant phenotypes for thousands of bacterial genes of unknown function." Nature 557:503–509. PMID: 29769716
- Deutschbauer A, Price MN, Wetmore KM, Shao W, Baumohl JK, Xu Z, Nguyen M, Tamse R, Davis RW, Arkin AP. (2011). "Evidence-based annotation of gene function in Shewanella oneidensis MR-1 using genome-wide fitness profiling across 121 conditions." PLoS Genetics 7:e1002385. PMID: 22125499
- Wetmore KM, Price MN, Waters RJ, Lamson JS, He J, Hoover CA, Blow MJ, Bristow J, Butland G, Arkin AP, Deutschbauer A. (2015). "Rapid quantification of mutant fitness in diverse bacteria by sequencing randomly bar-coded transposons." mBio 6:e00306-15. PMID: 25968644
- Price MN, Deutschbauer AM, Arkin AP. (2024). "A comprehensive update to the Fitness Browser." mSystems 9:e00470-24.
- Vaccaro BJ, Lancaster WA, Thorgersen MP, Zane GM, Younkin AD, Kazakov AE, Wetmore KM, Deutschbauer A, Arkin AP, Novichkov PS, Wall JD, Adams MW. (2016). "Novel Metal Cation Resistance Systems from Mutant Fitness Analysis of Denitrifying Pseudomonas stutzeri." Appl Environ Microbiol 82:6046–6056. PMID: 27474723
- Makarova KS, Wolf YI, Koonin EV. (2019). "Towards functional characterization of archaeal genomic dark matter." Biochem Soc Trans 47:389–398. PMID: 30647141
- Arkin AP, Cottingham RW, Henry CS, Harris NL, Stevens RL, Masber S, et al. (2018). "KBase: The United States Department of Energy Systems Biology Knowledgebase." Nature Biotechnology 36:566–569. PMID: 29979655
- Peters JM, Colavin A, Shi H, Czarny TL, Larson MH, Wong S, Hawkins JS, Lu CHS, Koo BM, Marta E, et al. (2016). "A Comprehensive, CRISPR-based Functional Analysis of Essential Genes in Bacteria." Cell 165:1493–1506. PMID: 27238023
- Peters JM, Koo BM, Patidar R, Heber CC, Tekin S, Cao K, Terber K, Lanze CE, Sirothia IR, Murray HJ, et al. (2019). "Enabling genetic analysis of diverse bacteria with Mobile-CRISPRi." Nature Microbiology 4:244–250. PMID: 30617347
- Tan SZ, Reisch CR, Prather KLJ. (2018). "A robust CRISPRi gene repression system in Pseudomonas." Journal of Bacteriology 200:e00575-17. PMID: 29311275
- Mimee M, Tucker AC, Voigt CA, Lu TK. (2015). "Programming a Human Commensal Bacterium, Bacteroides thetaiotaomicron, to Sense and Respond to Stimuli in the Murine Gut Microbiota." Cell Systems 1:62–71. PMID: 26918244
- Pavlopoulos GA, Baltoumas FA, Liu S, Noval Rivas M, Pinto-Cardoso S, et al. (2023). "Unraveling the functional dark matter through global metagenomics." Nature 622:594–602. PMID: 37821698
- Zhang Y, Bhosle A, Bae S, Franzosa EA, Huttenhower C, et al. (2025). "Predicting functions of uncharacterized gene products from microbial communities." Nature Biotechnology. PMID: 41094150
- Alam MT, Takano E, Breitling R. (2011). "Prioritizing orphan proteins for further study using phylogenomics and gene expression profiles in Streptomyces coelicolor." BMC Research Notes 4:325. PMID: 21899768
- Koo BM, Kritikos G, Farelli JD, Todor H, Tong K, Kimber H, Wapinski I, Galardini M, Caber A, Peters JM, et al. (2017). "Construction and Analysis of Two Genome-Scale Deletion Libraries for Bacillus subtilis." Cell Systems 4:291–305. PMID: 28189581
- Dembek M, Barquist L, Boinett CJ, Cain AK, Mayho M, Lawley TD, Fairweather NF, Fagan RP. (2015). "High-throughput analysis of gene essentiality and sporulation in Clostridium difficile." mBio 6:e02383-14. PMID: 25714712
- de Vries SP, et al. (2017). "Genome-wide fitness analyses of the foodborne pathogen Campylobacter jejuni in in vitro and in vivo models." Scientific Reports 7:1251. PMID: 28455506
- Sastry AV, Gao Y, Szubin R, Hefner Y, Xu S, Kim D, Choudhary KS, Yang L, King ZA, Palsson BO. (2019). "The Escherichia coli transcriptome mostly consists of independently regulated modules." Nature Communications 10:5536. PMID: 31797920
Experimental Recommendations
This section distills the entire analysis into an actionable experimental campaign. Two complementary prioritization routes were developed — one optimized for genes with the strongest existing evidence, the other for maximizing discovery of completely unknown functions — and both converge on a tractable set of organisms.
Two complementary prioritization routes
Route A — Evidence-weighted prioritization (NB05–NB09): Scores each dark gene across 6 dimensions (fitness importance 0.25, conservation 0.20, inference 0.20, pangenome 0.15, biogeographic 0.10, tractability 0.10) using data available from the 48 Fitness Browser organisms. This route identifies genes where multiple lines of evidence converge on a testable hypothesis. Best for: targeted experiments on genes where we already know what to test and under what conditions.
Route B — Conservation-weighted prioritization (NB11, NB11b): Queries the full 27,690-species GTDB r214 pangenome to measure taxonomic breadth of each dark gene OG (species range 1–27,482), classifies by taxonomic tier (kingdom through species) and hypothesis status (strong hypothesis, weak lead, true knowledge gap), then ranks by importance = conservation × ignorance. This route identifies broadly conserved genes where we know the least. Best for: discovery experiments targeting the most fundamentally important unknowns in biology.
The two routes produce different organism orderings because they optimize different objectives:
| Route A (Evidence-weighted) | Route B (Conservation-weighted) | |
|---|---|---|
| Top organism | MR-1 (deep condition coverage, many converging evidence lines) | S. meliloti (1,630 OGs, 195 kingdom-level gaps) |
| Optimization | Σ(composite_score) × tractability × phylo_diversity | Σ(importance) × tractability × phylo_bonus |
| Organisms selected | 42 (28 genera), 95% of composite priority | 42 (28 genera), 95.6% of importance-weighted priority |
| Overlap | 39 organisms shared; Route A uniquely selects 3 (Dda3937, Ddia6719, Miya), Route B uniquely selects 3 (Kang, Methanococcus_JJ, Methanococcus_S2) | |
| Key strength | Condition-specific experimental protocols; testable hypotheses | Identifies broadly conserved true knowledge gaps invisible to evidence-based scoring |
| Best for | Targeted RB-TnSeq under predicted conditions | Broad phenotypic screens for fundamentally unknown genes |
The most valuable dark genes and why
Of 57,011 dark genes across 48 bacteria, 17,344 have experimentally measurable phenotypes — either strong fitness defects (|fit| ≥ 2 in at least one condition) or essentiality (no viable transposon mutants). These are not computationally predicted to matter; they demonstrably matter in the lab. Among them, the convergence of multiple independent evidence lines (fitness phenotype, conservation, co-regulation, domain structure, gene neighborhood, cross-organism concordance) identifies a subset that are both important and interpretable.
Route A top fitness-active candidates — genes where fitness data, module co-regulation, domain annotations, and biogeographic signals converge to produce testable functional hypotheses:
| Rank | Gene | Organism | Score | Hypothesis | Top Condition | Why Valuable |
|---|---|---|---|---|---|---|
| 1 | AO356_11255 | P. putida N2C3 | 0.715 | D-alanyl-D-alanine carboxypeptidase (EC 3.4.16.4) | nitrogen (fit=3.4) | Module prediction + EamA domain + strongest biogeographic signal (lab-field OR=44) + NMDC nitrogen correlation |
| 2 | 202463 | S. oneidensis MR-1 | 0.698 | Stress-responsive membrane protein (PF01145) | stress (fit=6.4) | Highest fitness magnitude among top candidates + YGGT domain + co-regulated module |
| 3 | 199738 | S. oneidensis MR-1 | 0.698 | K03306 family (nitrogen metabolism) | nitrogen (fit=5.5) | Part of a three-gene paralog family (with 203545, 202450) — comparing mutants tests functional redundancy |
| 4 | 203545 | S. oneidensis MR-1 | 0.694 | K03306 family (DUF4124 domain) | nitrogen (fit=4.0) | Paralog of 199738; same module, different domain — suggests subfunctionalization |
| 5 | 202450 | S. oneidensis MR-1 | 0.693 | K03306 family (Gly_transporter domain) | nitrogen (fit=3.9) | Third K03306 paralog; glycine transporter domain suggests nitrogen/amino acid link |
Route B top knowledge gaps — the most broadly conserved genes with zero functional evidence, where experimental characterization would produce the most novel biological insight:
| Rank | Root OG | Species | Phyla | Tier | Hypothesis Status | Importance | Description |
|---|---|---|---|---|---|---|---|
| 1 | COG0468 | 27,427 | 142 | kingdom | true knowledge gap | 23.9 | Pan-bacterial, present in virtually all sequenced genomes |
| 2 | COG0443 | 27,279 | 142 | kingdom | true knowledge gap | 23.9 | Pan-bacterial, functionally uncharacterized |
| 3 | COG0491 | 27,393 | 142 | kingdom | true knowledge gap | 23.9 | Pan-bacterial, zero evidence across all inference layers |
These OGs are among the most universally conserved genes in bacteria yet remain functionally uncharacterized. They are invisible to Route A because they may lack condition-specific fitness effects (they may be essential under all conditions, producing no differential fitness signal).
The top essential dark gene candidates are genes where no viable knockout mutants exist, but gene neighborhood context, cross-species conservation, and domain structure enable CRISPRi knockdown experiments:
| Rank | Gene | Organism | Score | Domain | Evidence |
|---|---|---|---|---|---|
| 1 | 14796 | E. coli K-12 | 0.875 | YbeY | Conserved in 5+ phyla, synteny-confirmed operon, predicted ion transport |
| 2 | 200382 | S. oneidensis MR-1 | 0.874 | RimP_N/DUF150_C | Predicted ribosome assembly factor, conserved neighborhood |
| 3 | BWI76_RS08540 | K. oxytoca | 0.865 | OmpA/TIGR02802 | Predicted cell division, conserved across Enterobacteriaceae |
These candidates score highest because multiple independent evidence lines converge: they have detectable domains (providing structural clues), belong to widely conserved ortholog groups (confirming biological importance beyond one organism), sit in conserved gene neighborhoods with annotated partners (providing functional context), and are in organisms amenable to CRISPRi (making them experimentally testable despite being essential).
Which organisms and why
Route A organism selection (NB09): A greedy weighted set-cover algorithm selected 42 organisms (28 genera) covering 95% of the total priority value across all scored dark genes. The algorithm optimizes composite priority score × tractability × phylogenetic diversity.
1. Shewanella oneidensis MR-1 (587 genes, tractability 0.8) — Selected first because it combines deep condition coverage (121 conditions historically), a large dark gene complement spanning all tiers (172 T5 Dawn, 257 T4 Penumbra), and high genetic tractability. 544 of its dark genes have specific condition recommendations (stress, nitrogen, carbon). MR-1 is the single highest-impact organism: 25/100 top fitness-active candidates reside here.
2. Pseudomonas fluorescens N1B4 (624 genes, tractability 0.7) — Contributes the most additional uncovered genes after MR-1, particularly in carbon/nitrogen source panels (215 genes) and membrane stress (77 genes).
3. Sinorhizobium meliloti (570 genes, tractability 0.6) — Covers a distinct phylogenetic lineage (Alphaproteobacteria) with 316 T4 Penumbra and 90 T5 Dawn genes.
4. Escherichia coli K-12 (Keio) (368 genes, tractability 0.9) — Highest tractability; 259 hypothesis-bearing genes immediately testable; 205 essential genes ideal for CRISPRi.
5. Klebsiella oxytoca (396 genes, tractability 0.65) — Covers Enterobacteriaceae genes not in E. coli, including top essential candidate BWI76_RS08540 (score 0.865).
Route B organism selection (NB11): A conservation-weighted set-cover algorithm selected 42 organisms covering 95.6% of total importance-weighted priority. The algorithm optimizes Σ(importance) × tractability × phylo_bonus.
1. Sinorhizobium meliloti (1,630 OGs, 195 kingdom gaps, tractability 0.6) — Selected first because its dark genes span the most kingdom-level true knowledge gaps — broadly conserved genes with zero functional evidence.
2. Pseudomonas putida (1,043 OGs, 172 kingdom gaps, tractability 0.8) — High tractability combined with deep coverage of conserved unknowns.
3. Shewanella oneidensis MR-1 (805 OGs, 105 kingdom gaps, tractability 0.8) — Third in Route B vs. first in Route A, because Route B weights conservation breadth over condition-specific evidence depth.
4. Bacteroides thetaiotaomicron (1,382 OGs, 267 kingdom gaps, tractability 0.3) — Highest raw OG count of any organism; deprioritized by low tractability (0.3) but critical for non-Proteobacteria coverage (Bacteroidota).
5. Klebsiella michiganensis (568 OGs, 65 kingdom gaps, tractability 0.65) — Enterobacteriaceae depth beyond E. coli.
The first 10 Route B organisms cover 31% of importance-weighted priority (9,014 genes across 10 genera).
Phylogenetic gap analysis: beyond the 48 Fitness Browser organisms
Both covering sets draw only from the 48 FB organisms, which are heavily biased: 37/48 are Pseudomonadota (Proteobacteria), with Bacteroidota (4), Cyanobacteriota (1), and Halobacteriota (2) the only other phyla represented. Major bacterial phyla — Bacillota (Firmicutes), Actinomycetota (Actinobacteria), and Campylobacterota — have zero FB representation. This means kingdom-level OGs (55.9% of dark gene OGs, present across multiple phyla) cannot be experimentally addressed in their non-Proteobacterial hosts using FB organisms alone.
To address this gap, we curated an extended tractable organism list of 73 organisms (48 FB + 25 literature-curated with published TnSeq or CRISPRi resources; data/extended_tractable_organisms.tsv). The 25 non-FB organisms fill specific phylogenetic gaps:
| Phylum | Organisms added | Technology | Key references |
|---|---|---|---|
| Bacillota (6) | B. subtilis 168, S. aureus USA300, S. pneumoniae TIGR4, C. difficile R20291, E. faecium, L. monocytogenes EGD-e | TnSeq + CRISPRi | Koo et al. 2017; Bae et al. 2004; van Opijnen et al. 2009; Dembek et al. 2015 |
| Actinomycetota (2) | M. tuberculosis H37Rv, C. glutamicum ATCC 13032 | TnSeq + CRISPRi | DeJesus et al. 2017; Cleto et al. 2016 |
| Campylobacterota (2) | C. jejuni NCTC 11168, H. pylori 26695 | TnSeq | Gao et al. 2014; Salama et al. 2004 |
| Pseudomonadota (15) | P. aeruginosa PAO1, Salmonella 14028s, V. cholerae N16961, A. baumannii 17978, + 11 others | TnSeq/CRISPRi | Turner et al. 2015; Langridge et al. 2009; Chao et al. 2013 |
Incorporating these organisms into the Route B covering set would enable experimental characterization of kingdom-level dark gene OGs in their native Gram-positive, Actinobacterial, and Campylobacterial genomic contexts. This requires a Spark query to map each non-FB organism's genome to dark gene root_ogs via the pangenome — a natural extension of the NB11b pipeline.
How to study them: the experimental strategy
The action plan classifies each dark gene into one of two experimental categories:
Hypothesis-bearing genes (14,450 across all 42 organisms): These have at least one condition recommendation from fitness data, module co-regulation, or gene neighborhood context. The experiment is targeted RB-TnSeq or CRISPRi under the predicted condition. For example, MR-1:202463 (YGGT domain, stress-responsive) should be tested under oxidative, osmotic, and heat stress; the K03306 paralog trio (199738/203545/202450) should be tested under nitrogen limitation with single and double mutant comparisons.
Darkest genes requiring broad screens (2,038 across all 42 organisms): These are T1 Void or T2 Twilight genes with detectable fitness phenotypes but no converging evidence to suggest a specific condition. The experiment is a broad phenotypic screen — RB-TnSeq across a diverse condition panel (the standard Fitness Browser protocol of ~50-100 conditions). For E. coli K-12 alone, 109 genes fall in this category.
Essential genes (8,900 in covering set): These cannot be studied by transposon knockout. The recommended approach is CRISPRi knockdown (Mobile-CRISPRi for non-model organisms, Peters et al. 2019) with growth curves under standard and stress conditions. The top 50 essential candidates have specific CRISPRi experiment designs in NB07.
Prioritized entry points — a three-experiment starting campaign (Route A):
- MR-1 stress screen: RB-TnSeq under 5 stress conditions (oxidative, osmotic, metal, heat, pH). Addresses 161 hypothesis-bearing dark genes including the #2 overall candidate (202463, fit=6.4 under stress).
- MR-1 nitrogen screen: RB-TnSeq under nitrogen limitation and amino acid supplements. Addresses the K03306 paralog family (3 genes, fits 3.9–5.5) and 74 total dark genes.
- E. coli K-12 CRISPRi: Knockdown of top 20 essential dark genes with growth curves. Leverages the highest-tractability organism (0.9) and mature CRISPRi tools. Top target: Keio:14796 (YbeY domain, score 0.875).
Discovery campaign (Route B): For labs focused on novel function discovery rather than hypothesis testing, the Route B ordering — starting with S. meliloti (195 kingdom-level gaps), P. putida (172 kingdom gaps), and MR-1 (105 kingdom gaps) — maximizes the probability of uncovering completely new biology. Broad phenotypic screens under diverse condition panels are recommended for Route B organisms, since the target genes are true knowledge gaps without condition predictions.
These campaigns are complementary: Route A produces mechanistic insights for genes where we have hypotheses; Route B discovers functions for genes where we have none. The full action plans are in data/experimental_action_plan.tsv (Route A) and data/conservation_experiment_plans.tsv (Route B).
Discoveries
Full pangenome analysis (27,690 species, NB11b) reveals that over half of functionally dark gene OGs are pan-bacterial, present in thousands of species across multiple phyla. Species counts range from 1 to 27,482 (median 135). This demonstrates that functional dark matter is not a minor annotation g
Read more →The darkness spectrum classification across 57,011 dark genes shows T1 Void (zero evidence) is only 4,273 genes (7.5%). The majority — T4 Penumbra (22,500, 39.5%) — have 3-4 converging lines of evidence (fitness phenotype, domain annotation, ortholog group, module membership). The "dark matter probl
Read more →Across 47 testable dark gene clusters, 29 (61.7%) show directional concordance between lab fitness condition and carrier genome environment (Fisher's combined p=0.031). NMDC independent validation confirmed all 4 pre-registered abiotic predictions (nitrogen~nitrogen, pH~pH, anaerobic~dissolved_oxyge
Read more →65 dark gene ortholog groups show conserved fitness effects across 3+ organisms. Dark gene concordance levels are statistically indistinguishable from annotated genes (Mann-Whitney p=0.17). Motility-related dark genes show the strongest cross-organism concordance, suggesting conserved but incomplete
Read more →Shewanella MR-1 contributes 25/100 top fitness-active candidates, 587 scored dark genes total, and covers 20.8% of the top-500 in just 3 experiments (stress, nitrogen, carbon). This reflects deep condition coverage (121 conditions), a large dark gene complement, and high fitness effect magnitudes (1
Read more →Evidence-weighted (Route A, NB09) and conservation-weighted (Route B, NB11) covering sets both select 42 organisms covering ~95% of priority, sharing 39 organisms. Route A puts MR-1 first (deep condition evidence); Route B puts S. meliloti first (most kingdom-level gaps). The complementarity enables
Read more →Extended covering set spans 6 phyla but Bacillota OGs are subsets of Pseudomonadota coverage
February 2026Adding 25 non-FB organisms (Bacillota, Actinomycetota, Campylobacterota) to the covering set candidate pool selects 50 organisms spanning 6 phyla (vs 4 for FB-only). P. aeruginosa PAO1 ranks #1 (3,713 OGs) and M. tuberculosis reaches #6 (first Actinomycetota). However, Bacillota organisms (B. subtil
Read more →Data Collections
Used By
Data from this project is used by other projects.
Review
Summary
This is an exceptionally comprehensive project that catalogs 57,011 functionally dark genes across 48 bacteria, integrates evidence from four prior observatory projects and three BERDL collections, applies six novel inference layers (GapMind gap-filling, cross-organism concordance, phylogenetic breadth, biogeographic carrier analysis, lab-field concordance, NMDC validation), and produces a dual-route experimentally prioritized candidate list with organism-level covering sets. The project spans 13 notebooks (255 code cells, all with saved outputs), generates 39 figures, produces 44 data files, and is supported by thorough documentation including a detailed research plan with pre-registered hypotheses, a comprehensive 844-line report with 14 findings, and 12 explicitly stated limitations. The statistical methodology is sound throughout, with appropriate use of FDR correction, pre-registered predictions, stratified controls, sensitivity analysis, and robust rank indicators. Notably, many suggestions from the prior review cycle have been addressed — domain matching for GapMind (NB10), robust rank indicators (NB10), species-count scoring variant (NB10), formal binomial and sign tests (NB10), and full pangenome conservation (NB11b). The main remaining areas for improvement are a handful of code-level bugs (operator precedence in NB06, stale string constant in NB05, potential concordance score asymmetry), the mobile gene classification heuristic sensitivity, and the opportunity for a proper null comparison in the biogeographic analysis.
Methodology
Research question and hypotheses: The research question is clearly stated and decomposed into five testable sub-hypotheses (H1a–H1e). The null hypothesis is well-defined, and mixed outcomes are honestly anticipated. The pre-registration of condition-environment mappings before examining biogeographic data (NB04) is a commendable methodological safeguard rarely seen in computational biology projects.
Approach soundness: The multi-layered evidence integration is well-motivated. Each notebook addresses a question the preceding layer cannot answer: GapMind provides metabolic context beyond fitness data; concordance confirms cross-organism reproducibility; biogeography tests ecological relevance. The dual-route framework (Route A evidence-weighted, Route B conservation-weighted) is a thoughtful design that acknowledges different experimental objectives rather than forcing a single ranking. The 10-revision research plan documents the evolution of the project's design with intellectual honesty.
Data sources: Clearly identified across three BERDL collections (kescience_fitnessbrowser, kbase_ke_pangenome, nmdc_arkin) and four prior observatory projects. The RESEARCH_PLAN's "Tables Required" section lists 20 tables with estimated row counts and filter strategies — a model for reproducibility. The explicit table documenting what was loaded from prior work vs. newly derived prevents credit ambiguity.
Reproducibility: Strong. The README includes a Reproduction section specifying prerequisites, Spark vs. local dependencies per notebook, and execution order. A requirements.txt with 9 dependencies is present. All 13 notebooks have 100% code cells with saved outputs (255/255). The figures/ directory contains all 39 referenced figures (5.3 MB total). Every intermediate data file is saved to data/ (147 MB, 44 files), enabling downstream notebooks to run from cached results without re-running Spark queries. One gap: estimated runtimes per notebook are not provided, which would help users plan execution on BERDL JupyterHub.
Code Quality
SQL correctness and pitfall awareness: The notebooks demonstrate strong awareness of BERDL pitfalls documented in docs/pitfalls.md:
- Fitness Browser string columns are properly CAST to FLOAT/INT throughout (NB01: CAST(gf.fit AS FLOAT), CAST(gf.t AS FLOAT); NB06: CAST(s.minFit AS FLOAT), CAST(s.nInOG AS INT))
- Spark temp views used instead of large IN clauses (NB01 target_loci, NB03 target_species/target_clusters/target_genomes, NB06 target_annot_ogs, NB11b target_root_ogs with 11,774 OGs)
- BROADCAST hints applied for small lookup tables joined against billion-row tables (NB03 cell 13: /*+ BROADCAST(tc) */)
- GapMind MAX score aggregation with correct four-level hierarchy via CASE expression (NB02)
- ncbi_env EAV format properly pivoted before use (NB03)
- AlphaEarth 28% coverage reported with every biogeographic claim
- Essential genes handled as a separate scoring class (NB07) since they structurally cannot have genefitness rows
Statistical methods: Generally appropriate and well-chosen:
- Fisher's exact test for 2×2 contingency tables (NB03, NB04, NB06)
- Mann-Whitney U and KS tests for distribution comparisons (NB03, NB06)
- BH-FDR correction applied systematically throughout (NB03, NB04, NB06)
- Spearman rank correlation for scoring sensitivity and NMDC validation (NB05, NB10)
- Pre-registered vs. exploratory test separation with independent FDR correction (NB06)
- Stratified sampling of annotated control OGs matching dark OG organism-count distribution (NB06)
- Binomial and sign tests for concordance rate assessment (NB10)
- Fisher's combined probability for aggregate evidence across 47 individual tests (NB10)
Notebook organization: All 13 notebooks follow a consistent structure: markdown header → setup → data loading → analysis → visualization → save. All figures use plt.show() for inline rendering. Summary cells provide tabular overviews.
Bugs identified:
-
NB06 cell 6 — Operator precedence bug (moderate): The expression
dark['top_condition_class'].notna() & (dark['is_core'] == True) | (dark['is_auxiliary'] == True)evaluates as(notna() & is_core) | is_auxiliarydue to&binding tighter than|, including all auxiliary genes regardless of whether they have a condition class. This affects only the extended H1b analysis (cell 6), not the main H1b test (cells 4–5) which pre-filters correctly. The parenthesized intent should benotna() & ((is_core) | (is_auxiliary)). -
NB06 — Concordance score asymmetry (moderate): NB06 cell 13 identifies and fixes a bug where
n_strongcan exceedn_organismsin specog concordance computation and applies the fix to annotated control OGs. However, the dark gene concordance scores loaded from NB02'sconcordance_scores.tsvmay contain the same uncorrected bug. The Mann-Whitney comparison in cell 14 could compare corrected annotated scores against uncorrected dark scores. Since this bug tends to inflate concordance and dark genes already show median=1.0, the impact on the H1 conclusion (p=0.17) may be modest, but the asymmetry should be documented or resolved. -
NB05 cell 7 — Stale essentiality class string (minor):
score_fitness()checksessentiality_class == 'essential_all', but NB01 produces'universally_essential'. Theis_essential_darkboolean fallback covers the main case so the essentiality bonus (0.15) is still applied, but this branch is dead code. -
NB11b — Mobile gene classification heuristic (minor): The threshold
n_species / n_phyla <= 10for mobile element detection may be too aggressive. An OG present in 20 species across 2 phyla (ratio=10) would be classified as "mobile" even though this pattern is consistent with a genuinely conserved but phylum-sparse gene. The 6.5% mobile rate should be interpreted cautiously. -
NB03 — Embedding dimensionality reduction (minor): Mann-Whitney U on L2 norms of 64-dimensional embeddings reduces a multivariate comparison to univariate, potentially missing structured differences. A PERMANOVA would be more principled, though the current approach is reasonable given only 1/67 significant results.
-
NB01 cell 25 — Per-organism SQL loop (minor): Iterates 48 individual domain queries instead of a single query with a temp view. Functionally correct but inefficient.
Findings Assessment
Findings well-supported by data: The 14 findings follow directly from notebook analyses with specific numbers, tables, and figures. The REPORT's Results section provides narrative context explaining why each analysis step was needed — a welcome structural choice that transforms raw results into a scientific argument.
Strongest findings:
- Finding 1 (24.9% dark, 17,344 with phenotypes): Directly derived from data integration; aligns with published 25–40% estimates.
- Finding 4 (65 concordant OGs): Cross-organism concordance validated by NB06 null control (dark genes indistinguishable from annotated, p=0.17).
- Finding 7 (61.7% concordance + 4/4 NMDC confirmations): Lab-field concordance rate is now formally tested (binomial p=0.072, Fisher's combined p=0.031; NB10). The 7/7 NMDC trait sign test (p=0.0078) is the most convincing statistical result.
- Finding 12 (998 double-validated pairs): Synteny + co-fitness provides STRING-like evidence from Fitness Browser data alone. The explicit comparison against DOOR/STRING/EFI-GNT standards shows appropriate self-awareness.
- Finding 13 (darkness spectrum): The five-tier classification transforms "dark matter" from monolithic to actionable — only 7.5% truly lack all evidence.
- Finding 14 (full pangenome conservation): NB11b corrects the original species-count range from 1–33 to 1–27,482. The revelation that 55.9% of dark gene OGs are kingdom-level is a significant finding about the scale of the annotation gap.
Areas where prior review suggestions were addressed:
- Domain matching for GapMind (Suggestion 1 → NB10 Section 1: 42,239 candidates, 5,398 high-confidence EC matches)
- Robust rank indicators (Suggestion 2 → NB10 Section 2: 18 always-top-50 fitness, 6 always-top-50 essential)
- Species-count scoring variant (Suggestion 3 → NB10 Section 3: Spearman ρ=0.982, 62% top-50 overlap)
- Formal binomial test (Suggestion 6 → NB10 Section 5: p=0.072, Fisher's combined p=0.031)
Findings with caveats:
- Finding 3 (GapMind): NB10's domain matching substantially improves on organism-level co-occurrence, but the report correctly notes full gene-to-step validation requires AlphaFold or experimental enzymology.
- Finding 6 (10/137 significant biogeographic clusters): Modest hit rate. The P. putida clinical isolate enrichment for stress/nitrogen genes is ecologically interesting but not intuitive.
- Finding 5 (phylogenetic breadth): The eggNOG coarseness limitation is honestly reported and addressed by NB11b's full pangenome analysis.
Limitations: Comprehensively documented (12 items) covering environmental metadata sparsity, NMDC genus-level resolution, annotation bias, module prediction confidence, condition coverage unevenness, GapMind scope, essential gene scoring penalty, NMDC trait compositional coupling, missing biogeographic null, gene neighborhood methodology gaps, scoring weight sensitivity, and Proteobacteria organism bias. This level of self-criticism is exemplary and exceeds what most projects provide.
Suggestions
Important
-
Fix operator precedence in NB06 cell 6: Add parentheses to correctly filter the extended H1b analysis:
dark['top_condition_class'].notna() & ((dark['is_core'] == True) | (dark['is_auxiliary'] == True)). Re-run and verify results are unchanged or update accordingly. -
Reconcile concordance scores between NB02 and NB06: Either (a) retroactively apply the
nunique()fix from NB06 cell 13 to the dark gene concordance computation in NB02, regeneratingconcordance_scores.tsv, or (b) add a sentence in Limitation 9 noting that the dark-vs-annotated comparison may have an asymmetry in concordance score computation. -
Fix stale essentiality class string in NB05: Change
'essential_all'to'universally_essential'inscore_fitness(). The dead code branch is misleading and could cause bugs if scoring logic is reused. -
Add a biogeographic null control using annotated accessory genes: The report acknowledges this gap (Limitation 9). Running the NB03–NB04 pipeline on a matched set of annotated accessory genes (same organisms, same conservation status, known function) would test whether the 61.7% concordance rate exceeds what's expected for any accessory gene. With the marginal binomial p=0.072, this controlled comparison would substantially strengthen or appropriately weaken the H1d conclusion.
-
Document mobile gene heuristic sensitivity: Add a brief note in the REPORT's limitations or NB11b that the mobile detection threshold (
n_species / n_phyla <= 10) was not systematically optimized, and the 6.5% mobile rate may include genuinely conserved but phylum-sparse genes. Consider testing alternative thresholds (e.g., 5, 15) and reporting the range of mobile rates.
Nice-to-Have
-
Add estimated runtimes to the Reproduction section: For each Spark-dependent notebook, provide approximate wall-clock times on BERDL JupyterHub (e.g., "NB01: ~15 min, NB11b: ~5 min"). This helps users plan execution sessions.
-
Consolidate per-organism SQL loop in NB01: Replace the 48 individual domain queries (cell 25) with a single query using a temp view of organism IDs. Functionally equivalent but cleaner.
-
Add
low_memory=Falseto large TSV reads: NB11b shows a DtypeWarning from mixed-type columns indark_genes_integrated.tsv. Addinglow_memory=Falseto thosepd.read_csv()calls suppresses the warning. -
Version-pin dependencies more tightly: The
requirements.txtuses>=constraints. For long-term reproducibility, consider adding upper bounds (e.g.,pandas>=2.0,<3.0) or a lockfile.
This review was generated by an AI system. It should be treated as advisory input, not a definitive assessment.
Visualizations

Fig01 Annotation Breakdown

Fig02 Fitness Distributions

Fig03 Dark Gene Coverage

Fig04 Condition Classes

Fig05 Gapmind Gaps

Fig06 Concordance

Fig07 Phylo Breadth

Fig08 Env Distribution

Fig09 Carrier Tests

Fig10 Embedding Umap

Fig11 Concordance Matrix

Fig12 Nmdc Correlations

Fig13 Score Components

Fig14 Top20 Dossiers

Fig15 Organism Distribution

Fig16 H1B Test

Fig17 Concordance Comparison

Fig18 Neighbor Analysis

Fig19 Essential Scores

Fig20 Essential Top20

Fig21 Trait Correlations

Fig22 Conserved Neighborhoods

Fig23 Cofit Validation

Fig24 Improved Experimental Roadmap

Fig25 Darkness Spectrum

Fig26 Covering Set

Fig27 Action Plan

Fig28 Domain Matching

Fig29 Robust Ranks

Fig30 Species Count Scoring

Fig31 Statistical Tests

Fig32 Organism Taxonomy

Fig33 Bakta Reclassification

Fig33 Conservation Tiers

Fig34 Bakta Coverage Heatmap

Fig34 Classification Heatmap

Fig35 Top Knowledge Gaps

Fig36 Covering Set Curve

Fig37 Experiment Plan Heatmap

Fig38 Pangenome Species Distribution

Fig39 Extended Covering Set
Notebooks
01_integration_census.ipynb
01 Integration Census
View notebook →
02_gapmind_concordance_phylo.ipynb
02 Gapmind Concordance Phylo
View notebook →
03_biogeographic_analysis.ipynb
03 Biogeographic Analysis
View notebook →
04_lab_field_concordance.ipynb
04 Lab Field Concordance
View notebook →
05_prioritization_dossiers.ipynb
05 Prioritization Dossiers
View notebook →
06_robustness_checks.ipynb
06 Robustness Checks
View notebook →
07_essential_dark_prioritization.ipynb
07 Essential Dark Prioritization
View notebook →
08_improved_neighborhoods.ipynb
08 Improved Neighborhoods
View notebook →
09_final_synthesis.ipynb
09 Final Synthesis
View notebook →
10_review_improvements.ipynb
10 Review Improvements
View notebook →
11_conservation_classes.ipynb
11 Conservation Classes
View notebook →
11b_extended_conservation.ipynb
11B Extended Conservation
View notebook →
11c_extended_covering_set.ipynb
11C Extended Covering Set
View notebook →
12_bakta_enrichment.ipynb
12 Bakta Enrichment
View notebook →
Data Files
| Filename | Size |
|---|---|
biogeographic_profiles.tsv |
2.8 KB |
carrier_genome_map.tsv |
323.6 KB |
carrier_noncarrier_tests.tsv |
51.3 KB |
concordance_detailed.tsv |
3.8 KB |
concordance_scores.tsv |
2.5 KB |
conservation_covering_set.tsv |
4.7 KB |
conservation_experiment_plans.tsv |
7.8 KB |
dark_gene_census_full.tsv |
13778.5 KB |
dark_gene_classes.tsv |
7109.7 KB |
experimental_action_plan.tsv |
9.1 KB |
experimental_roadmap.tsv |
4.3 KB |
extended_covering_set.tsv |
7.3 KB |
extended_tractable_organisms.tsv |
10.0 KB |
gapmind_domain_matched.tsv |
4015.1 KB |
gapmind_gap_candidates.tsv |
55.5 KB |
gapmind_pathway_summary.tsv |
2.4 KB |
improved_candidates.tsv |
37.3 KB |
lab_field_concordance.tsv |
12.0 KB |
minimum_covering_set.tsv |
2578.1 KB |
nmdc_validation.tsv |
10.1 KB |
non_fb_genus_og_coverage.tsv |
1150.7 KB |
og_id_root_propagation.tsv |
117.6 KB |
og_importance_ranked.tsv |
1249.1 KB |
og_pangenome_distribution.tsv |
3221.6 KB |
og_pangenome_distribution_fb_only.tsv |
455.1 KB |
phylogenetic_breadth.tsv |
2842.8 KB |
prioritized_candidates.tsv |
81.8 KB |
robust_ranks_essential.tsv |
880.3 KB |
robust_ranks_fitness.tsv |
1645.4 KB |
scoring_all_dark.tsv |
3567.9 KB |
scoring_species_count_variant.tsv |
1764.8 KB |